| Hantavirus I, The Species and
The Rats invasion./
Hantavirus I, Las Especies y La invasion de
las Ratas.
DATA-MEDICOS
DERMAGIC/EXPRESS 19-(206)
24 Septiembre 2.017 24 September 2.017
 HANTAVIRUS II, Hemorrhagic Fever with Renal
Syndrome & Pulmonary Syndrome. /Fiebre Hemorragica con Sindrome
Renal y Sindrome Pulmonar.
(HOT LINK)
HANTAVIRUS II, Hemorrhagic Fever with Renal
Syndrome & Pulmonary Syndrome. /Fiebre Hemorragica con Sindrome
Renal y Sindrome Pulmonar.
(HOT LINK)
EDITORIAL ENGLISH
===================
Hello friends of the DERMAGIC EXPRESS network
today brings you a very interesting topic THE
HANTAVIRUS I, THE SPECIES AND THE RATS INVASION,
rodent borne zoonotic disease, a topic related
to the aforementioned
BUNYAVIRIDAE FAMILY virus. It is
impressive the number of SPECIES found to date
in relation to these HANTAVIRUS, which are
practically spread throughout the globe,
representing a mortality of 10 to 36% in those
affected.
Many of them are benign for the man, it means
that it has not been shown PATHOGENICITY or
ability to produce disease in the HUMAN, but
some of them produce the so-called
HEMORRHAGIC FEVER WITH RENAL SYNDROME (HFRS) or
EPIDEMIC NEPHROPATHY with a mortality of.10-12%
and
HANTAVIRUS PULMONARY OR CARDIOPULMONARY SYNDROME
(HPS) to which a mortality has been
described between 25-36% in some countries.
The name HANTA VIRUS comes from the
HANTAN RIVER area in
SOUTH KOREA, where it was discovered
by scientists in the late 1970s by the Korean
virologist Ho-Wang Lee. And the American Karl M.
Johnson.
Ho-Wang Lee described in Korea Seoul, The
SEOUL HANTAVIRUS (SEOV)
transmitted by rodents and causing the
HEMORRHAGIC FEVER HANTAVIRUS WITH RENAL
SYNDROME (HFRS) or EPIDEMIC NEPHROPATHY
for the first time in 1.976
The main carriers or reservoirs of these viruses
are
RODENTS: mice and rats through urine,
saliva, fluids and feces that are sprayed in the
environment can infect you. In some cases
bats European mole, shrews. And European common
vole are involved like hosts, but in the case
of these two diseases that I mentioned the main
natural reservoirs are
RODENTS.
The HANTAVIRUS belong to the
group of
VIRUS TYPE RNA,
order:
NOT ASSIGNED,
family
BUNYAVIRIDAE,
genus:
HANTAVIRUS, Type of
species:
HANTAAN VIRUS with more than
50 SUBTYPES OR SPECIES
which I place here and the country where
they were discovered or described by first time.
I believe that you will be
astonished of the number of HANTAVIRUS
SPECIES described until today 2,017 !!!!
THE HANTA VIRUS SPECIES DESCRIBED UNTIL TODAY
2.017
==================================================
1.)
Andes virus:
Argentina and
Chile; (1.997-1998)
2.)
Amur virus:
Far East of
Russia, China, and Korea. 2.002-2.004)
3.)
Asama virus:
Japan (2.008)
4.)
Azagny virus: west
Africa (2012).
5.)
Bayou virus: Southeastern
United States. (1944)
6.)
Black Creek Canal virus:
Black Creek Canal area of
Dade County, Florida in 1995.
7.)
Bloodland Lake virus: Pulaski
County,
Missouri in 1994.
8.)
Blue River virus:
Jackson County, Missouri in 1995.
9..)
Cano Delgadito virus: Caño
Delgadito virus is a Hantavirus. It was
discovered in the 1990s from rodent species in
Los Llanos in Venezuela.
10.)
Calabazo virus:
Central América on the Azuero
Peninsula of Panamá in early 2000.
11.)
Carrizal virus:
Guerrero,
Mexico.
2012
12.)
Catacamas virus:
Honduras 2.006
13.)
Choclo virus: 1999 in Western
Panama.
14.)
Dobrava-Belgrade
VIRUS: Found in
Dobrava Village, Slovenia, Yugoslavia It
was subsequently isolated in striped field mice
in
Russia and other parts of
Eastern Europe. It has also been found in
Germany but the reservoir host there is
unknown.
15.)
El Moro Canyon virus:
El Moro Canyon in
southeastern Colorado in 1995.
16.
Gou virus: Zhejiang Province in
2000.
China
16,)
Hantaan River virus:
Korea 1.976 date of war from years
1951-1.953 mortality 10-30%
17.)
Huitzilac virus: Morelos and
Guerrero,
Mexico. 2.012
18.)
Imjin virus:
Republic of
Korea during 2004 and 2005.
19.)
Isla Vista virus:
Santa Barbara County,
California 1.995
20.)
Khabarovsk virus:
Far-east
Russia. 1.996
21.)
Laguna Negra virus: South America
Brazil
22.)
Limestone Canyon virus:
The United States and
South America.
23.)
Magboi virus: Magboi Stream in
eastern
Sierra Leone in 2011
24.)
Maripa virus:
French Guiana 2.012
25.)
Monongahela virus: Monongahela
National Forest in
West Virginia in 1985
26.)
Montano virus:
Morelos and Guerrero,
Mexico. 2.012
27.)
Mouyassue virus:
Sierra Leone 2.012, Bats, share lineage
with Magboi virus
28.)
Muleshoe virus: Deaf Smith County,
Texas in 1995
29.)
Muju virus: Republic of
Korea. 2.007
30.)
New York virus:
New York 2.013
31.)
Nova virus:
Hungary and
France 2.014, no human infection has been
discovered (European mole and bats).
32.)
Oran virus:
South America related to
Andes virus
33.)
Oxbow virus: Gresham,
Oregon, in September 2003.
34.)
Playa de Oro viruS:
Mexican
state of Colima, 2.004 2.008
35.)
Prospect Hill virus:
meadow vole found in
Prospect Hill, Maryland for which it is
named. 1.982-1987
36.)
Puumala virus: in
Scandinavia and
Finland, although it has also been
reported elsewhere in
Northern Europe,
Poland and
Russia, 1.980
37,)
Rockport virus: Rockport,
Texas 2.011
38.)
Rio Mamore virus: Bolivia.
1.997
39.)
Rio Segundo virus:
Costa Rica, Related with
Sin Nombre virus (nameless)
40.)
Sangassou virus:
Guinea, West
Africa in 2010, bat, moles, and shrews.
41.)
Saaremaa virus: Found in striped
field mouse in
Slovakia. 2.006
42.)
Seoul virus: Seoul
Korea.
43.)
Serang virus: Serang,
Indonesia in 2.008.
44.)
Sin Nombre virus (no name): It was
first isolated in 1.993 from rodents collected
near the home of one of the initial patients
with HANTAVIRUS PULMONARY SYNDROME (HPS) in the
Four Corners region of the
western United States.
45.)
Soochong virus:
Korea August 1997
46.)
Tanganya virus
:
Africa 2.007
47.)
Thailand virus:
Thai provinces, Nakhon Pathom and Nakhon
Ratchasima, in
Thailand 1994
48.)
Thottapalayam virus:
India in 1964.
49.)
Topografov virus: Topografov River
in the Taymyr Peninsula,
Siberia. 1.996
50.)
Tula VIRUS:
first isolated from a European common vole (Microtus
arvalis) found in Central
Russia 1994
51.)
Xuan Son virus: Pomona
Vietnam 2.013
52.)
Seewis virus:
Germany
53.)
asikkala virus:
Germany
54.)
Araraquara virus:
Brazil
55.)
Juquitiba virus:
Brazil, possible lineage of the
ARARAQUARA VIRUS
56.)
Anajatuba Virus, Bermejo Virus,
Jabora Virus, Lechiguanas virus,
Maciel virus,
Oran virus, Pergamino virus,
Rio Mearim Virus: Strains of the
ANDES VIRUS:
Argentina, Chile, Brazil, Uruguay, Paraguay.
Summarizing the countries involved so far are:
Argentina, Chile, Bolivia, Honduras, Venezuela,
Panama, United States, Russia, China, Indonesia,
Siberian, Thailand, Slovakia, Germany, Hungary,
France, Poland, Korea, Vietnam, India, Mexico,
Costa Rica, Scandinavia, Finland, Brazil, French
Guiana, Japan, Africa and others
Other countries where
species of
HANTAVIRUS have been found:
Holland, Bulgaria, Bosnia and Herzegovina,
Paraguay, Uruguay, England, Wales, Belgium,
Sweden, Canada and Colombia.
It is estimated that
in China between 1950 and 2007
there were 1.5 million cases per
HANTAVIRUS, of
which 46,000 died,
in Russia there were 90,000, cases
from 1996 to 2006,
in Finland 32,000, between
2005 and 2010.
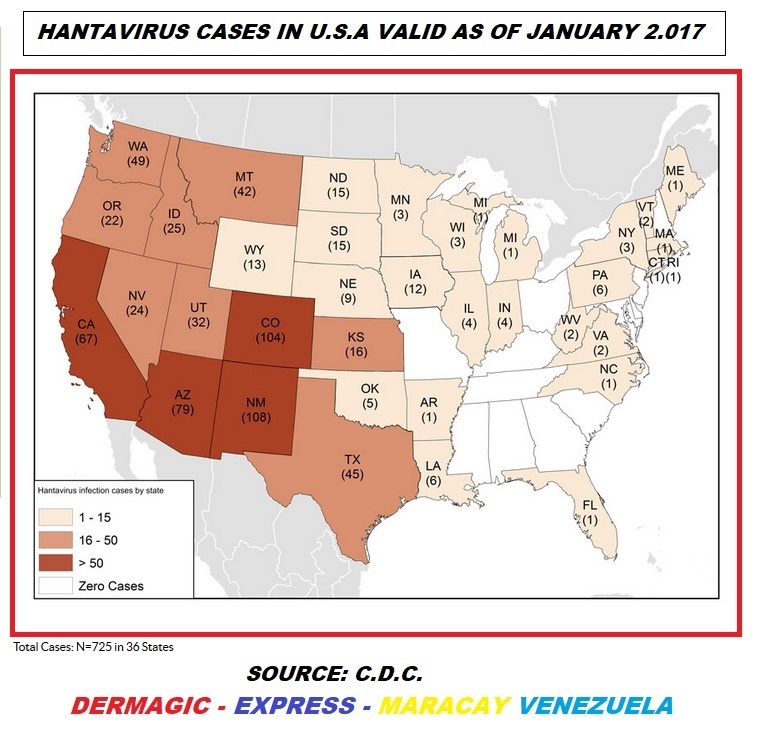
In the United States up to January
2,017, 725 cases were reported, of which
697 occurred in the first outbreak in 1993,
USA Hantavirus mortality
averaged 36%.
In French Guiana between 2008 and
2016, 5 cases of HANTAVIRUS PULMONARY
SYNDROME (HPS) were reported from the MARIPA
VIRUS, of which
4 died, 80% mortality.
In Argentina in an outbreak of
20 cases of PULMONARY SYNDROME (HPS)
by HANTAVIRUS in 1997
the HUMAN-HUMAN transmission was demonstrated
by the
virus ANDES (ANDV).
And here comes the following reflection:
rodents, rats and mice known ancestrally
from Ancient Rome and perhaps before, are
spread throughout the PLANET and are
transmitters of more than 26 diseases
among which stand out
BUBONIC PLAGUE (Yerinia Pestis),
LEPTOSPIROSIS , SALMONELLOSIS, TAENIASIS,
TOXOPLASMOSIS, TRICHINOSIS AND FEVER.
HANTAVIRUS IS DISSEMINATED THROUGH these
rodents all over the planet, some of them LETAL,
in the next edition I will talk
about the DISEASES, RODENTS and VIRUSES
involved.
Greetings to all
Dr. Jose Lapenta.
EDITORIAL ESPAÑOL
=================
Hola amigos de la red DERMAGIC EXPRESS te
trae hoy un tema bastante interesante LOS
HANTAVIRUS I, LAS ESPECIES Y LA INVASION DE
LAS RATAS, zoonosis transmitida por
roedores, tema relacionado con los ya
mencionados virus de la
FAMILIA BUNYAVIRIDAE. Es
impresionante la cantidad de ESPECIES
encontradas hasta el día de hoy en relación
a estos HANTA VIRUS, los cuales están
prácticamente diseminados por todo el globo
terráqueo, representando una mortalidad
del 10 al 36% en los afectados.
Muchos de ellos son benignos para el hombre,
quiere decir que no se ha demostrado
PATOGENICIDAD o capacidad de producir
enfermedad en el HUMANO, pero algunos de
ellos producen la llamada
FIEBRE HEMORRAGICA CON SINDROME RENAL (HFRS)
o NEFROPATIA EPIDEMICA con una
mortalidad del 10 al 12% y
el SINDROME PULMONAR O CARDIOPULMONAR POR
HANTAVIRUS (HPS). al cual se le
ha descrito una mortalidad entre 25 y 35%
en algunas regiones.
El nombre de
HANTA VIRUS
proviene del área del
RIO DE HANTAN en
COREA DEL SUR, donde fue
descubierto por los científicos a finales de
los años 70 por el virologista Coreano
Ho-Wang Lee. y Karl M. Johnson.
Ho-Wang Lee describio el
SEOUL HANTVIRUS (SEOV)
transmitido por roedores y causante de la
FIEBRE HEMORRAGICA HANTAVIRUS CON
SINDROME RENAL (HFRS) o NEFROPATIA EPIDEMICA
por primera vez.
Los principales portadores o reservorios de
estos virus son los
ROEDORES: ratones y ratas a través de
orina, saliva, fluidos y heces que
pulverizadas en el ambiente pueden
contagiarte. En algunos casos están
involucrados murciélagos, tapir, musarañas
y topillo campesino. pero en el
caso de estas dos enfermedades que les
mencione los reservorios principales
naturales son
ROEDORES.
Los
HANTAVIRUS pertenecen al
grupo
de
VIRUS TIPO ARN,
orden:
NO ASIGNADO, familia BUNYAVIRIDAE,
genus: HANTAVIRUS, Tipo de especie: HANTAAN
VIRUS con
más de 50 SUBTIPOS O especies
las cuales te coloco aquí y el país donde
fueron descubiertos o descritos por primera
vez.
Creo que te quedaras
asombrado
del número de ESPECIES DE
HANTAVIRUS descritas hasta hoy día 2.017
LAS ESPECIES DE HANTA VIRUS DESCRITAS
HASTA HOY DIA 2.017:
=========================================================
1.)
Andes virus:
Argentina y Chile; (1.997-1998)
2.)
Amur virus:
Corea
y
china noreste, lejano Oriente de
Rusia, 2.002-2.004)
3.)
Asama virus:
Japón (2.008)
4.)
Azagny virus:
África del este: (2012).
5.)
Bayou virus:
Sureste de
Estados unidos. (1944)
6.)
Black Creek Canal virus: Black
Creek Canal área del
Condado Dade, Florida en 1995.
7.)
Bloodland Lake virus:
Pulaski Condado,
Missouri en 1994.
8.)
Blue River virus: Condado
de Jackson, Missouri en 1995.
9.)
Cano Delgadito virus:
Caño Delgadito virus es un
hantavirus. Descubierto en los años
90 en roedores en
Los Llanos en Venezuela.
10.)
Calabazo virus:
America
Central en la Península de Azuero
Panamá en los años 2000.
11.)
Carrizal VIRUS: Guerrero,
México. 2012
12.)
Catacamas virus:
Honduras
2.006
13.)
Choclo virus:
1999 en el Oeste
de
Panamá.
14.)
Dobrava-Belgrade
VIRUS: en la población de
Dobrava, Eslovenia, Yugoslavia.
Subsecuentemente fue aislado en
Rusia en el Ratón Rayado de Campo y
otras partes del
Este de Europa. También fue
encontrado en
Alemania pero el reservorio en este
país se desconoce.
15.)
El Moro Canyon virus: El Cañón
Moro en el
Sureste de Colorado en 1995.
16.
Gou virus:
En la provincia de
Zhejiang
China en el 2000.
16,)
Hantaan River virus:
Corea 1.976, data de la guerra años
1951-1.953 mortalidad 10-30%
17.)
Huitzilac virus: Morelos y
Guerrero,
México. 2.012
18.)
Imjin virus:
Republica de
Corea años 2004 y 2005.
19.)
Isla Vista virus:
Condado de
Santa Barbará, California 1.995
20.)
Khabarovsk virus: Lejano
Oriente
Rusia. 1.996
21.)
Laguna Negra virus:
Sur
América
Brasil
22.)
Limestone Canyon virus:
Estados Unidos y Suramérica.
23.)
Magboi virus: Magboi Stream en
el Este de
Sierra Leona en 2011
24.)
Maripa virus:
Guayana Francesa 2.012
25.)
Monongahela virus:
Monongahela
Bosque Nacional en el
Oeste de Virginia en 1985
26.)
Montano virus:
Morelos y Guerrero,
México. 2.012
27.)
Mouyassue virus:
Sierra Leona
2.012 Murciélago,
comparte Linaje con Magboi virus.
28.)
Muleshoe virus: Condado Deaf
Smith,
Texas en 1995
29.)
Muju VIRUS: Republica DE
Corea.
2.007
30.)
New York virus:
New York 2.013
31.)
Nova virus:
Hungría y
Francia 2.014 No se ha descrito
infección en humanos (Tapir y Murciélagos).
32.)
Oran virus:
Sur América
relacionado con el
Andes virus
33.)
Oxbow virus: Gresham,
Oregón, en Septiembre del 2003.
34.)
Playa de Oro viruS: Estado
de
Colima México, 2.004 2.008
35.)
Prospect Hill virus:
Prospect Hill, Maryland del cual
proviene su nombre. 1.982-1987
36.)
Puumala virus:
Escandinavia y
Finlandia, 1980, aunque también ha
sido reportado en cualquier parte
de Norte de Europa, Polonia
y
Rusia.
37,)
Rockport virus: Rockport,
Texas
2.011
38.)
Rio Mamore virus: Bolivia.
1.997
39.)
Rio Segundo virus:
Costa Rica, Relacionado con el
Sin Nombre virus
40.)
Sangassou virus:
Guinea, Oeste
de
África en 2010. Murciélagos, Tapir y
musarañas.
41.)
Saaremaa virus: Ratón rayado
de campo en
Eslovaquia. 2.006
42.)
Seoul virus:
Seúl
Corea.
43.)
Serang virus:
Serang,
Indonesia in 2008.
44.)
Sin Nombre virus: Fue aislado
por primera vez en 1993 de roedores en la
región de
Las Cuatro Esquinas
en el
Oeste de Los Estados Unidos en
pacientes con el SINDROME PULMONAR POR
HANTAVIRUS (HPS).
45.)
Soochong virus:
Corea Agosto 1997
46.)
Tanganya VIRUS:
África 2.007
47.)
Thailand virus:
Provincia de Thai, Nakhon Pathom y Nakhon
Ratchasima,
Tailandia
en 1994
48.)
Thottapalayam virus:
India in 1964.
49.)
Topografov virus:
Topografov,
rio en la Península de Taymyr Península,
Siberia.
1.996
50.)
Tula virus:
Aislado por primera vez en
Europa del Topillo campesino
(Microtus arvalis) encontrado en
Rusia Central en 1994
51.)
Xuan Son virus:
Pomona
Vietnam
2.013
52.)
Seewis virus:
Alemania
53.)
asikkala virus:
Alemania.
54.)
Araraquara virus:
Brasil
55.)
Juquitiba virus:
Brasil, posible linaje del
ARARAQUARA VIRUS
56.)
Anajatuba Virus, Bermejo Virus,
Jabora Virus, Lechiguanas virus,
Maciel virus,
Oran virus, Pergamino virus,
Rio Mearim Virus: Cepas del
ANDES VIRUS:
Argentina, Chile, Brasil, Uruguay, Paraguay.
Resumiendo los países hasta ahora
involucrados son:
Argentina, Chile, Bolivia, Honduras,
Venezuela, Panamá, Estados Unidos, Rusia,
China, Indonesia, Siberia, Tailandia,
Eslovaquia, Alemania, Hungría, Francia,
Polonia, Corea, Vietnam, India, México,
Costa Rica, Escandinavia, Finlandia, Brasil,
Guayana Francesa, Japón, África y otros
Otros países donde se ha encontrado
especies de
HANTAVIRUS:
Holanda, Bulgaria, Bosnia y Herzegovina,
Paraguay, Uruguay, Inglaterra, Gales,
Bélgica, Suecia Canadá y Colombia.
Se calcula que
en China entre 1.950 y 2.007
hubo 1.5 millones de casos por
HANTAVIRUS, de los cuales
murieron 46.000,
en Rusia hubo 90.000 mil
casos desde 1.996 hasta 2.006 ,
en Finlandia 32.000 mil
entre 2.005 y 2.010.
En Estados Unidos hasta Enero
de 2.017 se reportaron 725 casos de
los cuales 697 ocurrieron en el primer brote
en 1.993, la mortalidad en USA por
Hantavirus
promedia el 36%
En la Guayana Francesa
entre 2.008 y 2016 se reportaron 5 casos
de SINDROME PULMONAR POR HANTAVIRUS (HPS)
por el VIRUS MARIPA,
de los cuales 4 murieron,
mortalidad 80%
En Argentina en un brote de 20
casos de SINDROME PULMONAR(HPS) por
HANTAVIRUS en 1.997 se demostro la
transmision HUMANO-HUMANO, por el virus
ANDES (ANDV).
Y aquí viene la siguiente reflexión:
los roedores , ratas y ratones
conocidos ancestralmente desde la Antigua Roma y
quizá antes, están diseminados por todo EL
PLANETA y son transmisores de más de 26
enfermedades entre las que destacan la
PESTE BUBONICA (yersinia Pestis),
LEPTOSPIROSIS, SALMONELOSIS, TENIASIS,
TOXOPLASMOSIS, TRIQUINOSIS Y FIEBRE.
Los HANTAVIRUS ESTAN DISEMINADOS A TRAVES de
estos roedores en todo el planeta,
algunos de ellos
LETALES, en la
próxima edición TE hablare de las
ENFERMEDADES, ROEDORES y VIRUS involucrados
Saludos a Todos.
Dr. José Lapenta.
=======================================================================
REFERENCIAS
BIBLIOGRAFICAS/
BIBLIOGRAPHICAL REFERENCES
=======================================================================
1.) Country-wide seroprevalence studies
on Crimean-Congo hemorrhagic fever and
hantavirus
infections in general population of
Bulgaria.
2.) Serological Survey of Zoonotic
Viruses in Invasive and Native Commensal
Rodents in Senegal, West Africa.
3.) Seoul hantavirus in brown rats in
the Netherlands: implications for
physicians--Epidemiology, clinical
aspects, treatment and diagnostics.
4.) Co-circulation of Araraquara and
Juquitiba Hantavirus in Brazilian
Cerrado.
5.) Serogrouping and seroepidemiology
of North European hantaviruses using a
novel broadly targeted synthetic
nucleoprotein antigen array.
6.) [Hantaviruses in Germany: threat
for zoo, pet, companion and farm animals?].
7.) Hantavirus infections in Europe and
their impact on public health.
8.) Detection of different South
American hantaviruses.
9.) Spatial prediction and validation
of zoonotic hazard through micro-habitat
properties: where does Puumala
hantavirus hole - up?
10.) Hantaviruses and their hosts in
Europe: reservoirs here and there, but
not everywhere?
11.) Habitat factors associated with
bank voles (Clethrionomys glareolus) and
concomitant hantavirus in northern
Sweden.
12.) [Increase in the number of cases
of epidemic nephropathy in Germany.
Virological and
ecological aspects].
13.) A comparative epidemiological
study of hantavirus infection in Japan
and Far East Russia.
14.) Puumala virus outbreak in Western
Thuringia, Germany, 2010: epidemiology
and strain identification.
15.) Environmental conditions in favour
of a hantavirus outbreak in 2015 in
Germany?
16.) Hantavirus Pulmonary Syndrome
Caused by Maripa Virus in French Guiana,
2008–2016.
17.) An unusual hantavirus outbreak in
southern Argentina: person-to-person
transmission? Hantavirus Pulmonary
Syndrome Study Group for Patagonia.
==================================================================
======================================================================
1.) Country-wide seroprevalence studies
on Crimean-Congo hemorrhagic fever and
hantavirus
infections in general population of
Bulgaria.
=======================================================================
J Med Virol. 2017 Oct;89(10):1720-1725.
doi: 10.1002/jmv.24868. Epub 2017 Jul 6.
Christova I1, Panayotova E1, Trifonova
I1, Taseva E1, Hristova T1, Ivanova V1.
Author information
1
National Center of Infectious and
Parasitic Diseases, Sofia, Bulgaria.
Abstract
Crimean-Congo hemorrhagic fever (CCHF)
and hantavirus infections are the two
viral hemorrhagic fevers spread in
Europe. To test actual circulation of
CCHF virus (CCHFV) and hantaviruses in
Bulgaria, we conducted country-wide
seroepidemiological studies. Serum
samples were collected prospectively
from 1500 residents of all 28 districts
in Bulgaria. CCHFV seroprevalence of
3.7% was revealed. Anamnesis for tick
bites, contact with livestock, age over
40 years and residency in Haskovo
district were found as risk factors. The
highest CCHFV seroprevalence was
observed in the known endemic districts
in southeastern Bulgaria: Haskovo (28%)
and Yambol (12%). Reactive samples were
found in residents of 20 of the 28
districts in Bulgaria. In comparison
with the previous studies, the data
presented indicate that CCHFV increased
substantially its circulation in the
endemic regions and was introduced in
many new areas. Hantavirus
seroprevalence was based on results of
the immunoblot and estimated as 3.1%.
Surprisingly, contrary to all available
data, Puumala virus seroprevalence rate
was 2.3% versus 0.8% of Dobrava-Belgrade
virus. Evidence for hantavirus IgG
seropositivity was found in residents of
23 of the 28 districts in the country.
The first hantavirus seroprevalence
study in Bulgaria showed that Puumala
virus is probably more wide-spread in
the country than Dobrava-Belgrade virus.
======================================================================
2.) Serological Survey of Zoonotic
Viruses in Invasive and Native Commensal
Rodents in Senegal, West Africa.
=======================================================================
Vector Borne Zoonotic Dis. 2017 Sep 5.
doi: 10.1089/vbz.2017.2135. [Epub ahead
of print]
Diagne CA1,2,3, Charbonnel N4,
Henttonen H5, Sironen T6, Brouat C1.
Author information
1
1 CBGP, IRD, CIRAD, INRA, Montpellier
SupAgro, University of Montpellier ,
Montpellier, France .
2
2 BIOPASS (IRD-CBGP, ISRA, UCAD),
Dakar, Senegal .
3
3 Département de Biologie Animale,
Faculté des Sciences et Techniques,
Université Cheikh Anta Diop (UCAD) ,
Dakar, Senegal .
4
4 CBGP, INRA, CIRAD, IRD, Montpellier
SupAgro, University of Montpellier ,
Montpellier, France .
5
5 Forest and Animal Ecology, Natural
Resources Institute Finland , Helsinki,
Finland .
6
6 Department of Virology, University of
Helsinki , Helsinki, Finland .
Abstract
Increasing studies on rodent-borne
diseases still highlight the major role
of rodents as reservoirs of numerous
zoonoses of which the frequency is
likely to increase worldwide as a result
of accelerated anthropogenic changes,
including biological invasions. Such a
situation makes pathogen detection in
rodent populations important, especially
in the context of developing countries
characterized by high infectious disease
burden. Here, we used indirect
fluorescent antibody tests to describe
the circulation of potentially zoonotic
viruses in both invasive (Mus musculus
domesticus and Rattus rattus) and native
(Mastomys erythroleucus and Mastomys
natalensis) murine rodent populations in
Senegal (West Africa). Of the 672
rodents tested, we reported 22
seropositive tests for Hantavirus,
Orthopoxvirus, and Mammarenavirus
genera, and no evidence of viral
coinfection. This study is the first to
report serological detection of
Orthopoxvirus in rodents from Senegal,
Mammarenavirus in R. rattus from Africa,
and Hantavirus in M. m. domesticus and
in M. erythroleucus. Further specific
identification of the viral agents
highlighted here is urgently needed for
crucial public health concerns.
======================================================================
3.) Seoul hantavirus in brown rats in
the Netherlands: implications for
physicians--Epidemiology, clinical
aspects, treatment and diagnostics.
=======================================================================
Neth J Med. 2015 May;73(4):155-60.
Goeijenbier M1, Verner-Carlsson J, van
Gorp EC, Rockx B, Koopmans MP, Lundkvist
Å, van der Giessen JW, Reusken CB.
Author information
1
Department of Virology, Erasmus MC,
Rotterdam, the Netherlands.
Abstract
The recent discovery of Seoul
hantavirus (SEOV) presence in wild rat
populations in the Netherlands has
direct implications for Dutch clinicians
and hantavirus diagnostics. SEOV is
amongst the Old World hantaviruses which
cause haemorrhagic fever and renal
syndrome (HFRS) in humans. HFRS is
characterised by a classical triad of
fever, acute kidney injury and
haemorrhage, but can show different
signs and symptoms in specific cases.
SEOV is transmitted from infected rats
to humans by inhalation of aerosolised
excreta. When compared with the known
circulating hantaviruses in the
Netherlands, Puumala (PUUV) and Tula (TULV),
SEOV causes a more severe form of HFRS.
Data from cohort studies undertaken in
China and Northern Europe show
differences in signs and symptoms at
onset of disease, (haemorrhagic)
complications and mortality. Furthermore,
routine diagnostics currently available
for hantavirus diagnosis in the
Netherlands are not optimised for SEOV
detection. The clinical outcome of an
SEOV and PUUV infection will greatly
benefit from an early diagnosis which
will reduce the costs of unnecessary
tests and treatments as well. The
discovery of SEOV circulation in the
Netherlands follows recent findings of
SEOV infections in both rodents and
humans in England, Wales, France,
Belgium and Sweden, indicating the
emerging character of SEOV and a high
importance of this hantavirus for Public
Health in large areas of Europe. Here,
we review the current knowledge on the
clinical manifestation of SEOV versus
PUUV infections in humans, the treatment
of clinical cases and diagnostics.
======================================================================
4.) Co-circulation of Araraquara and
Juquitiba Hantavirus in Brazilian
Cerrado.
======================================================================
Microb Ecol. 2017 Aug 31. doi:
10.1007/s00248-017-1061-4. [Epub ahead
of print]
Guterres A1,2, de Oliveira RC3,
Fernandes J3, Maia RM3, Teixeira BR4,
Oliveira FCG5, Bonvicino CR4,6, D'Andrea
PS4, Schrago CG7, de Lemos ERS8.
Author information
1
Laboratório de Hantaviroses e
Rickettsioses, Instituto Oswaldo Cruz,
Fundação Oswaldo Cruz, Pavilhão Helio e
Peggy Pereira - 1° Pav. Sala B115, Av.
Brasil 4365, Manguinhos, Rio de Janeiro,
RJ, 21045-900, Brazil. [email protected].
2
Departamento de Genética, Universidade
Federal do Rio de Janeiro, Rio de
Janeiro, Brazil. [email protected].
3
Laboratório de Hantaviroses e
Rickettsioses, Instituto Oswaldo Cruz,
Fundação Oswaldo Cruz, Pavilhão Helio e
Peggy Pereira - 1° Pav. Sala B115, Av.
Brasil 4365, Manguinhos, Rio de Janeiro,
RJ, 21045-900, Brazil.
4
Laboratorio de Biologia e Parasitologia
de Mamíferos Silvestres Reservatórios,
Instituto Oswaldo Cruz, Fundação Oswaldo
Cruz, Rio de Janeiro, RJ, Brazil.
5
Centro Tecnológico de Engenharia LTDA,
Goiânia, GO, Brazil.
6
Instituto Nacional do Câncer - INCA,
Rio de Janeiro, RJ, Brazil.
7
Departamento de Genética, Universidade
Federal do Rio de Janeiro, Rio de
Janeiro, Brazil.
8
Laboratório de Hantaviroses e
Rickettsioses, Instituto Oswaldo Cruz,
Fundação Oswaldo Cruz, Pavilhão Helio e
Peggy Pereira - 1° Pav. Sala B115, Av.
Brasil 4365, Manguinhos, Rio de Janeiro,
RJ, 21045-900, Brazil. [email protected].
Abstract
Hantavirus cardiopulmonary syndrome is
an emerging serious disease in the
Americas, transmitted from wild rodents
to humans through inhalation of aerosol
containing virus. Herein, we
characterized two distinct hantaviruses
circulating in rodent species form
Central Plateau, Midwestern region of
Brazil in the Cerrado (savanna-like)
biome, an area characterized by small
trees and grasses adapted to climates
with long dry periods. In this study, we
identified the co-circulation of the
Araraquara virus and a possible new
lineage of the Juquitiba virus (JUQV) in
Oligoryzomys nigripes. The implications
of co-circulation are still unknown, but
it can be the key for increasing viral
diversity or emergence of new species
through spillover or host switching
events leading to co-infection and
consequently recombination or
reassortment between different virus
species. Phylogenetic analyses based on
the complete S segment indicated that,
alongside with Oligoryzomys mattogrossae
rodents, O. nigripes species could also
have a whole as JUQV reservoir in the
Cerrado biome. Although these rodents'
species are common in the Cerrado biome,
they are not abundant demonstrating how
complex and different hantavirus
enzootic cycles can be in this
particular biome.
======================================================================
5.) Serogrouping and seroepidemiology
of North European hantaviruses using a
novel broadly targeted synthetic
nucleoprotein antigen array.
======================================================================
Infect Ecol Epidemiol. 2017 Jul
26;7(1):1350086. doi:
10.1080/20008686.2017.1350086.
eCollection 2017.
Rönnberg B1,2,3, Vapalahti O4,
Goeijenbier M5, Reusken C5, Gustafsson
Å3, Blomberg J1,2, Lundkvist Å1,2,3.
Author information
1
Section of Clinical Microbiology,
Department of Medical Sciences, Uppsala
University, Uppsala, Sweden.
2
Zoonosis Science Center, Department of
Medical Biochemistry and Microbiology,
Uppsala University, Uppsala, Sweden.
3
Laboratory of Clinical Microbiology,
Uppsala University Hospital, Uppsala,
Sweden.
4
Department of Veterinary Biosciences
and Virology, University of Helsinki and
Helsinki University Hospital, Helsinki,
Finland.
5
Department of Viroscience, Erasmus MC,
Rotterdam, The Netherlands.
Abstract
Introduction: Hantaviruses are globally
distributed zoonotic pathogens. Great
diversity and high antigenic cross-reactivity
makes diagnosis by traditional methods
cumbersome. Materials and methods: 'Megapeptides',
119-120-mers from the amino terminus of
the nucleoprotein of 16 hantaviruses,
representing the four major branches of
the hantavirus phylogenetic tree, were
utilized in a novel IgG-based hantavirus
suspension multiplex immunoassay (HSMIA)
for detection of past hantavirus
infections in 155 North European human
samples. We compared HSMIA with
established EIAs and focus reduction
neutralization test (FRNT). Results and
discussion: The Puumala hantavirus (PUUV)
component in the HSMIA gave concordant
results with a PUUV IgG EIA in 142 sera
from Northern Sweden (of which 31 were
EIA positive, 7 borderline and 104 EIA
negative, sensitivity 30/31 = 97%,
specificity 104/ 104 = 100%, 134/135 =
99% concordance), with another
immunoassay in 40 PUUV IgG positive sera
from Finland (36/40 = 90% sensitivity),
and was concordant in 8 of 11 cases with
PUUV and DOBV neutralization titers,
respectively. Two major IgG reactivity
patterns were found: (i) a PUUV-specific
pattern covering phylogroup IV and its
serogroups B and C; and (ii) a Dobrava
virus (DOBV)-specific pattern, covering
the serogroup A portion of phylogroup
III. In addition, we found several minor
patterns with reactivity to only one or
two megapeptides indicating additional
hantaviruses infecting humans in the
Swedish and Finnish populations.
Conclusion: The broadly reactive and
rational HSMIA yielded results highly
correlated with the established PUUV
EIAs and the NT results. It is a
sensitive and specific assay, which will
be suited for efficient serosurveillance
of hantaviruses in humans. Its use in
animals should be further investigated.
======================================================================
6.) [Hantaviruses in Germany: threat
for zoo, pet, companion and farm animals?].
======================================================================
Berl Munch Tierarztl Wochenschr. 2013
Nov-Dec;126(11-12):514-26.
[Article in German]
Ulrich RG1, Imholt C2, Krüger DH3,
Krautkrämer E2, Scheibe T4, Essbauer
SS5, Pfeffer M6.
Author information
1
Friedrich-Loeffler-Institut, Institut
für neue und neuartige
Tierseuchenerreger, Greifswald - Insel
Riems, Deutschland. [email protected]
2
Friedrich-Loeffler-Institut, Institut
für neue und neuartige
Tierseuchenerreger, Greifswald - Insel
Riems, Deutschland.
3
Nationales Konsiliarlaboratorium für
Hantaviren, Institut für Medizinische
Virologie, Helmut-Ruska-Haus, Charité -
Universitätsmedizin Berlin und
Fachbereich Virologie, Labor Berlin
Charité Vivantes GmbH.
4
Zoo Osnabrück, Osnabrück, Deutschland.
5
Institut für Mikrobiologie der
Bundeswehr, München, Deutschland.
6
Institut fŭr Tierhygiene und
Offentliches Veterinärwesen, Leipzig,
Deutschland.
Abstract
Hantaviruses are so-called "emerging"
and "re-emerging" viruses because of the
new and sudden nature of their
appearance. Human infections can lead to
two distinct disease patterns, the
Haemorrhagic Fever with Renal Syndrome
and the Hantavirus Cardiopulmonary
Syndrome. All known human pathogenic
hantaviruses are transmitted through
rodent hosts. There are three rodent-associated
hantaviruses in Germany. The bank vole-associated
Puumala virus (PUUV) is responsible for
most of the human hantavirus infections.
The Dobrava-Belgrade virus (DOBV)
associated with the striped field mouse
is causing hantavirus disease in the
North and Northeast of Germany. The
human pathogenicity of Tula virus (TULV)
is still controversially discussed--the
virus has been mainly associated with
the common vole as the reservoir, but
was molecularly detected also in the
field and the water vole. More recently,
two shrew-borne hantaviruses were
described in Germany, i. e. Seewis virus
in the common shrew and Asikkala virus
in the pygmy shrew. Systematic studies
about hantavirus infections of zoo, pet,
companion and farm animals are still
lacking. Hence, the aim of this review
article is to summarise the current
knowledge on this topic and raise the
attention of veterinarians to
potentially overlooked clinical disease
patterns.
======================================================================
7.) Hantavirus infections in Europe and
their impact on public health.
======================================================================
Rev Med Virol. 2013 Jan;23(1):35-49.
doi: 10.1002/rmv.1722. Epub 2012 Jul 3.
Vaheri A1, Henttonen H, Voutilainen L,
Mustonen J, Sironen T, Vapalahti O.
Author information
1
Department of Virology, Haartman
Institute, and Research Programs Unit,
Infection Biology, University of
Helsinki, Helsinki, Finland.
[email protected]
Abstract
Hantaviruses (genus Hantavirus, family
Bunyaviridae) are enveloped tri-segmented
negative-stranded RNA viruses each
carried by a specific rodent or
insectivore host species. Several
different hantaviruses known to infect
humans circulate in Europe. The most
common is Puumala (PUUV) carried by the
bank vole; another two important,
genetically closely related ones are
Dobrava-Belgrade (DOBV) and Saaremaa
viruses (SAAV) carried by Apodemus mice
(species names follow the International
Committee on Taxonomy of Viruses
nomenclature). Of the two hantaviral
diseases, hemorrhagic fever with renal
syndrome (HFRS) and hantaviral
cardiopulmonary syndrome, the European
viruses cause only HFRS: DOBV with often
severe symptoms and a high case fatality
rate, and PUUV and SAAV more often mild
disease. More than 10,000 HFRS cases are
diagnosed annually in Europe and in
increasing numbers. Whether this is
because of increasing recognition by the
medical community or due to
environmental factors such as climate
change, or both, is not known.
Nevertheless, in large areas of Europe,
the population has a considerable
seroprevalence but only relatively few
HFRS cases are reported. Moreover, no
epidemiological data are available from
many countries. We know now that cardiac,
pulmonary, ocular and hormonal disorders
are, besides renal changes, common
during the acute stage of PUUV and DOBV
infection. About 5% of hospitalized PUUV
and 16%-48% of DOBV patients require
dialysis and some prolonged intensive-care
treatment. Although PUUV-HFRS has a low
case fatality rate, complications and
long-term hormonal, renal, and
cardiovascular consequences commonly
occur. No vaccine or specific therapy is
in general use in Europe. We conclude
that hantaviruses have a significant
impact on public health in Europe.
======================================================================
8.) Detection of different South
American hantaviruses.
======================================================================
Guterres A1, de Oliveira RC2, Fernandes
J2, Schrago CG3, de Lemos ER2.
Author information
1
Laboratório de Hantaviroses e
Rickettsioses, Instituto Oswaldo Cruz,
Fundação Oswaldo Cruz, Rio de Janeiro,
RJ, Brazil; Departamento de Genética,
Universidade Federal do Rio de Janeiro,
Rio de Janeiro, Brazil. Electronic
address: [email protected].
2
Laboratório de Hantaviroses e
Rickettsioses, Instituto Oswaldo Cruz,
Fundação Oswaldo Cruz, Rio de Janeiro,
RJ, Brazil.
3
Departamento de Genética, Universidade
Federal do Rio de Janeiro, Rio de
Janeiro, Brazil.
Abstract
Hantaviruses are the etiologic agents
of Hemorrhagic Fever with Renal Syndrome
(HFRS) in Old World, and Hantavirus
Pulmonary Syndrome (HPS)/Hantavirus
Cardiopulmonary Syndrome (HCPS), in the
New World. Serological methods are the
most common approach used for laboratory
diagnosis of HCPS, however theses
methods do not allow the
characterization of viral genotypes. The
polymerase chain reaction (PCR) has been
extensively used for diagnosis of viral
infections, including those caused by
hantaviruses, enabling detection of few
target sequence copies in the sample.
However, most studies proposed methods
of PCR with species-specific primers.
This study developed a simple and
reliable diagnostic system by RT-PCR for
different hantavirus detection. Using
new primers set, we evaluated human and
rodent hantavirus positive samples of
various regions from Brazil. Besides, we
performed computational analyzes to
evaluate the detection of other South
American hantaviruses. The diagnostic
system by PCR proved to be a sensible
and simple assay, allowing amplification
of Juquitiba virus, Araraquara virus,
Laguna Negra virus, Rio Mamore virus and
Jabora virus, beyond of the possibility
of the detecting Andes, Anajatuba,
Bermejo, Choclo, Cano Delgadito,
Lechiguanas, Maciel, Oran, Pergamino and
Rio Mearim viruses. The primers sets
designed in this study can detect
hantaviruses from almost all known
genetics lineages in Brazil and from
others South America countries and also
increases the possibility to detect new
hantaviruses. These primers could easily
be used both in diagnosis of suspected
hantavirus infections in humans and also
in studies with animals reservoirs.
======================================================================
9.) Spatial prediction and validation
of zoonotic hazard through micro-habitat
properties: where does Puumala
hantavirus hole - up?
======================================================================
BMC Infect Dis. 2017 Jul 26;17(1):523.
doi: 10.1186/s12879-017-2618-z.
Khalil H1, Olsson G2, Magnusson M2,
Evander M3, Hörnfeldt B2, Ecke F2,4.
Author information
1
Department of Wildlife, Fish, and
Environmental Studies, Swedish
University of Agricultural Sciences,
Skogmarksgränd, 901 83, Umeå, Sweden.
[email protected].
2
Department of Wildlife, Fish, and
Environmental Studies, Swedish
University of Agricultural Sciences,
Skogmarksgränd, 901 83, Umeå, Sweden.
3
Department of Clinical Microbiology,
Virology, Umeå University, 901 85, Umeå,
Sweden.
4
Department of Aquatic Sciences and
Assessment, Swedish University of
Agricultural Sciences, Gerda Nilssons
väg 5, 756 51, Uppsala, Sweden.
Abstract
BACKGROUND:
To predict the risk of infectious
diseases originating in wildlife, it is
important to identify habitats that
allow the co-occurrence of pathogens and
their hosts. Puumala hantavirus (PUUV)
is a directly-transmitted RNA virus that
causes hemorrhagic fever in humans, and
is carried and transmitted by the bank
vole (Myodes glareolus). In northern
Sweden, bank voles undergo 3-4 year
population cycles, during which their
spatial distribution varies greatly.
METHODS:
We used boosted regression trees; a
technique inspired by machine learning,
on a 10 - year time-series (fall
2003-2013) to develop a spatial
predictive model assessing seasonal PUUV
hazard using micro-habitat variables in
a landscape heavily modified by forestry.
We validated the models in an
independent study area approx. 200 km
away by predicting seasonal presence of
infected bank voles in a five-year-period
(2007-2010 and 2015).
RESULTS:
The distribution of PUUV-infected voles
varied seasonally and inter-annually. In
spring, micro-habitat variables related
to cover and food availability in
forests predicted both bank vole and
infected bank vole presence. In fall,
the presence of PUUV-infected voles was
generally restricted to spruce forests
where cover was abundant, despite the
broad landscape distribution of bank
voles in general. We hypothesize that
the discrepancy in distribution between
infected and uninfected hosts in fall,
was related to higher survival of PUUV
and/or PUUV-infected voles in the
environment, especially where cover is
plentiful.
CONCLUSIONS:
Moist and mesic old spruce forests,
with abundant cover such as large holes
and bilberry shrubs, also providing food,
were most likely to harbor infected bank
voles. The models developed using long-term
and spatially extensive data can be
extrapolated to other areas in northern
Fennoscandia. To predict the hazard of
directly transmitted zoonoses in areas
with unknown risk status, models based
on micro-habitat variables and developed
through machine learning techniques in
well-studied systems, could be used.
======================================================================
10.) Hantaviruses and their hosts in
Europe: reservoirs here and there, but
not everywhere?
======================================================================
Vector Borne Zoonotic Dis. 2010 Aug;10(6):549-61.
doi: 10.1089/vbz.2009.0138.
Olsson GE1, Leirs H, Henttonen H.
Author information
1
Department of Wildlife, Fish, and
Environmental Studies, Swedish
University of Agricultural Sciences,
Umeå, Sweden. [email protected]
Abstract
Five hantaviruses are known to
circulate among rodents in Europe, and
at least two among insectivores. Four (Dobrava,
Saaremaa, Seoul, and Puumala [PUUV]
viruses) are clearly associated with
hemorrhagic fever with renal syndrome (HFRS).
PUUV, the most common etiological agent
of HFRS in Europe, is carried by the
bank vole (Myodes glareolus), one of the
most widespread and abundant mammal
species in Europe. This host-virus
system is among hantaviruses also the
most studied one in Europe. However,
HFRS incidence varies throughout the
continent. The spatial as well as
temporal variation in the occurrence of
HFRS is linked to geographic differences
in the population dynamics of the
reservoir rodents in different biomes of
Europe. While rodent abundance may
follow mast seeding events in many parts
of temperate Europe, in northern (N)
Europe multiannual cycles in population
density exist as the result of the
interaction between rodent populations
and specialist predator populations in a
delayed density-dependent manner. The
spatial distribution of hantaviruses
further depends on parameters such as
forest patch size and connectivity of
the most suitable rodent habitats, and
the conditions for the survival of the
virus outside the host, as well as
historical distribution patterns (phylogeographies)
of hosts and viruses. In multiannually
fluctuating populations of rodents, with
population increases of great amplitude,
one should expect a simultaneous build-up
of recently hantavirus-infected (shedding)
rodents. The increasing number of
infectious, virus-shedding rodents leads
to a rapid transmission of hantavirus
across the rodent population, and to
humans. Our review discusses these
aspects for PUUV, the only European
hantavirus for which there is a
reasonable, yet still far from complete,
ecological continental-wide
understanding. We discuss how this
information could translate to other
European hantavirus-host systems, and
where the most important questions lie
for further research.
======================================================================
11.) Habitat factors associated with
bank voles (Clethrionomys glareolus) and
concomitant hantavirus in northern
Sweden.
======================================================================
Vector Borne Zoonotic Dis. 2005 Winter;5(4):315-23.
Olsson GE1, White N, Hjältén J, Ahlm C.
Author information
1
Department of Animal Ecology, Swedish
University of Agricultural Sciences,
Umeå, Sweden. [email protected]
Abstract
Puumala virus (PUUV), genus hantavirus,
causes nephropathia epidemica, a mild
form of hemorrhagic fever with renal
syndrome in humans. In this study, bank
voles, the natural reservoir of PUUV,
were captured at locations of previous
human PUUV exposure and paired controls
within a region of high incidence in
northern Sweden. The aim of the study
was to evaluate the influence of
environmental factors on the abundance
of bank voles and the occurrence of PUUV.
The total number of voles and the number
of PUUV-infected voles did not differ
between locations of previous human PUUV
exposure and paired controls. The number
of bank voles expressing antibodies to
PUUV infection increased linearly with
total bank vole abundance implying
density independent transmission. Using
principal component and partial
correlation analysis, we found that
particular environmental characteristics
associated with old-growth moist forests
(i.e., those dominated by Alectoria
spp., Picea abies, fallen wood, and
Vaccinium myrtillus) were also
associated with increased abundance of
bank vole and hence the number of PUUV-infected
bank voles, whereas there were no
correlations with factors associated
with dry environments (i.e., Pinus
sylvestris and V. vitis-idea). This
suggests that circulation and
persistence of PUUV within bank vole
populations was influenced by habitat
factors. Future modeling of risk of
exposure to hantavirus and transmission
of PUUV within vole populations should
include the influence of these factors.
======================================================================
12.) [Increase in the number of cases
of epidemic nephropathy in Germany.
Virological and
ecological aspects].
======================================================================
Dtsch Med Wochenschr. 2008
Mar;133(10):476-8. doi:
10.1055/s-2008-1046736.
[Article in German]
Krautkrämer E1, Zeier M.
Author information
1
Nephrologie, Universitätsklinikum
Heidelberg, Im Neuenheimer Feld 162,
69120 Heidelberg. [email protected]
Abstract
Epidemic nephropathy (EN) is
transmitted to humans via rodents. The
causative agent of this virus-borne
renal disease is the Hantavirus Puumala.
Other members of the genus Hantavirus
cause hemorrhagic fever with renal
syndrome (HFRS) or hantaviral pulmonary
syndrome (HPS). As with all zoonoses the
prevalence of the virus depends on the
distribution of the reservoir species.
Climate changes have direct impact on
the number of host animals and influence
the incidence of hantaviral infections.
A number of studies demonstrate the
epidemiological relationship between
climate, food supply, rodent population
and outbreaks of HFRS and HPS. In
Germany the number of cases of EN has
increased in the past few years and huge
rise in the incidence of the infection,
more than 1600 cases, occurred in 2007.
======================================================================
13.) A comparative epidemiological
study of hantavirus infection in Japan
and Far East Russia.
======================================================================
Jpn J Vet Res. 2007 Feb;54(4):145-61.
Kariwa H1, Lokugamage K, Lokugamage N,
Miyamoto H, Yoshii K, Nakauchi M,
Yoshimatsu K, Arikawa J, Ivanov LI,
Iwasaki T, Takashima I.
Author information
1
Laboratory of Public Health, Graduate
School of Veterinary Medicine, Hokkaido
University, Sapporo, Japan. [email protected]
Abstract
Hantaviruses are causative agents of
some severe human illnesses, including
hemorrhagic fever with renal syndrome (HFRS)
and hantavirus pulmonary syndrome (HPS).
The viruses are maintained by rodent
hosts, and humans acquire infection by
inhaling virus-contaminated excreta from
infected animals. To examine the
epidemiology of hantavirus infections in
Japan and Far East Russia, we conducted
epidemiological surveys in these regions.
In Japan, anti-hantavirus antibodies
were found in four rodent species,
Clethrionomys rufocanus, Rattus
norvegicus, R. rattus, and Apodemus
speciosus. Although no new HFRS cases
have been officially reported over the
past 20 years in Japan, one member of
the Japan Ground Self-Defense Force did
test positive for hantavirus antibody.
Repeated surveys in Far East Russia have
revealed that two distinct hantavirus
types cause severe HFRS in this region.
Hantavirus sequences identified from A.
peninsulae, fetal HFRS cases in
Vladivostok, and Amur virus are highly
similar to each other (> 92% identity),
but they are less similar (approximately
84% identity) to the prototypical
Hantaan virus, which is carried by A.
agrarius. Phylogenetic analysis also
indicates that Amur and A. peninsulae-associated
viruses are distinct from Hantaan virus,
suggesting that A. peninsulae is the
reservoir animal for Amur virus, which
causes severe HFRS. From HFRS patients
in the Khabarovsk region, we identified
viruses with nucleotide sequences that
are more similar to Far East virus (>
96%identity) than to the Hantaan (88-89%
identity) or Amur (81-83% identity)
viruses. Phylogenetic analysis also
indicates that the viruses from
Khabarovsk HFRS patients are closely
related to the Far East virus, and
distinct from Amur virus.
======================================================================
14.) Puumala virus outbreak in Western
Thuringia, Germany, 2010: epidemiology
and strain identification.
======================================================================
Zoonoses Public Health. 2013 Dec;60(8):549-54.
doi: 10.1111/zph.12037. Epub 2013 Feb 8.
Faber M1, Wollny T, Schlegel M, Wanka
KM, Thiel J, Frank C, Rimek D, Ulrich RG,
Stark K.
Author information
1
Department for Infectious Disease
Epidemiology, Robert Koch Institute,
Berlin, Germany.
Abstract
In 2010, the highest annual number of
human Puumala virus (PUUV) infections
was reported in Germany since hantavirus
surveillance started in 2001. The
increase in annual case numbers was
especially marked in western Thuringia.
We combined results of case-based
hantavirus surveillance in humans and
serological and molecular investigations
in the rodent reservoir to describe the
epidemiological situation and to
identify the putative outbreak strain. A
5-fold increase in notified hantavirus
cases compared to the previous annual
maximum was observed in western
Thuringia in 2010. Disease incidence
varied tremendously within a small
geographical area with case patients'
places of residence clustering around
beech-dominated broad leaf forest
patches. Investigations in the rodent
reservoir revealed a novel Puumala virus
(PUUV) subtype, which is clearly
distinct from strains collected in other
PUUV endemic regions of Germany. It can
be assumed that in regions in western
Thuringia where hantavirus cases
occurred in 2010 or previous outbreak
years, PUUV has been present in the
environment for a long time. Further
studies are needed to elucidate the
population dynamics and hantavirus
prevalence of the rodent reservoir and
driving ecological factors.
======================================================================
15.) Environmental conditions in favour
of a hantavirus outbreak in 2015 in
Germany?
=======================================================================
Zoonoses Public Health. 2016
Mar;63(2):83-8. doi: 10.1111/zph.12217.
Epub 2015 Jul 15.
Reil D1,2, Imholt C1, Drewes S3, Ulrich
RG3, Eccard JA2, Jacob J1.
Author information
1
Julius Kühn-Institute, Institute for
Plant Protection in Horticulture and
Forests, Münster, Germany.
2
University of Potsdam, Institute of
Biochemistry and Biology, Potsdam,
Germany.
3
Friedrich-Loeffler-Institut, Institute
for Novel and Emerging Infectious
Diseases, Greifswald - Insel Riems,
Germany.
Abstract
Bank voles can harbour Puumala virus (PUUV)
and vole populations usually peak in
years after beech mast. A beech mast
occurred in 2014 and a predictive model
indicates high vole abundance in 2015.
This pattern is similar to the years
2009/2011 when beech mast occurred, bank
voles multiplied and human PUUV
infections increased a year later. Given
similar environmental conditions in
2014/2015, increased risk of human PUUV
infections in 2015 is likely. Risk
management measures are recommended.
================================================================
16.) Hantavirus Pulmonary Syndrome
Caused by Maripa Virus in French Guiana,
2008–2016
===============================================================
S. Matheus et al.
View Abstract
We report 5 human cases of hantavirus
pulmonary syndrome found during
surveillance in French Guiana in
2008–2016; of the 5 patients, 4 died.
This pathogen should continue to be
monitored in humans and rodents in
effort to reduce the occurrence of these
lethal l infections in humans stemming
from ecosystem disturbances.
==================================================================
17.) An unusual hantavirus outbreak in
southern Argentina: person-to-person
transmission? Hantavirus Pulmonary
Syndrome Study Group for Patagonia.
=================================================================
Emerg Infect Dis. 1997 Apr-Jun;3(2):171-4.
Wells RM1, Sosa Estani S, Yadon ZE,
Enria D, Padula P, Pini N, Mills JN,
Peters CJ, Segura EL.
Author information
1
Centers For Disease Control and
Prevention, Atlanta, Georgia, USA.
Abstract
Hantavirus pulmonary syndrome is a
rodent-borne zoonosis first recognized
in the United States in 1993. Person-to-person
transmission has not been reported;
however, in the outbreak of 20 cases
reported here, epidemiologic evidence
strongly suggests this route of
transmission.
=======================================================================
===================================================================
DATA-MEDICOS/DERMAGIC-EXPRESS No 19-(206) 24/09/2.017 DR. JOSE LAPENTA R.
===================================================================
Produced
by Dr. Jose Lapenta R. Dermatologist 2.017
Maracay Estado Aragua Venezuela 2.017
Telf: 0416-6401045- 02432327287-02432328571
loading...
|