| Marburg Virus, the night of
the bats. !!
Marburg Virus, la noche de los
Murcielagos !!
DATA-MEDICOS
DERMAGIC/EXPRESS 19-(204)
08 Octubre 2.017 08 October 2.017

EDITORIAL ENGLISH
==================
Hello friends of the DERMAGIC EXPRESS network today
brings you a very interesting topic, it is THE
MARBURG VIRUS (MARV), THE NIGHT OF BATS , virus
today considered
ONE OF THE MOST LETHAL VIRUSES for HUMANITY,
after the
EBOLA VIRUS, although both belong to the
same family of FILOVIRUS.
But perhaps the most
AMAZING I found in this review is that
this virus in the years of the COLD WAR in the
SOVIET UNION was one of the many biological
agents that I try to be used as a
biological weapon, to be placed in the
nuclear warheads of the Russian missiles.
This fact was denounced by the Russian scientist
Ken Alibek, who worked in one of the
laboratories in the years 1.983-1.988 for
such purposes and desert to the United States of
America later and revealed these state secrets.
Another detail to consider if you read previous
reviews, is that the habitat where humans live
is
NOT SO INOFENSIVE, we are surrounded
by
TICKS
(Lyme disease, Powassan, Heartland, Crimea-Congo
fever
and others)
INSECTS
(Zika, Dengue , Chikungunya, Nile Fever, Japanese
Encephalitis, jamestown canyon virus and
others),
RATS, MICE
(Hantavirus, Bubonic Plague, Leptospirosis,
Salmonella and others) virus
TRANSMITTERS of diseases that are
highly LETHAL or inoculate you with a
disease that may leave you sick lifelong . And today
I bring you also these,
THE BATS, harmless FLYERS MAMMALS that
live in caves, and nocturnal activity, NATURAL
RESERVOIRS OF THE VIRUSES OF MARBURG and EBOLA
considered today the 2
MOST LETHAL viruses for humanity with
mortality greater than 80%. After the
contagion.
THE MARBURG VIRUS (MARV) belongs to the
group:
RNA VIRUS, order:
MONONEGAVIRALES, family:
FILOVIRIDAE, genus:
MARBURGVIRUS, species:
MARGBURG MARBURGVIRUS.

MARBURG VIRUS appeared for the first time in
1.967 in a LABORATORY in the localities of
MARBURG and
FRANKFURT in
Germany and in
Belgrade,
Yugoslavia (today Serbia), when workers
were infected by an unknown pathogen that provoked
HEMORRHAGIC manifestations and high lethality.
The causal agent was isolated and identified by
scientists from the
University of MARBURG, Germany hence its name.
The virus carriers were infected
AFRICAN GREEN MONKEYS (Chlorocebus
aethiops) that had been imported to be used in the
manufacture of vaccines in the German HOECHST AG,
nowadays SANOFI-AVENTIS.
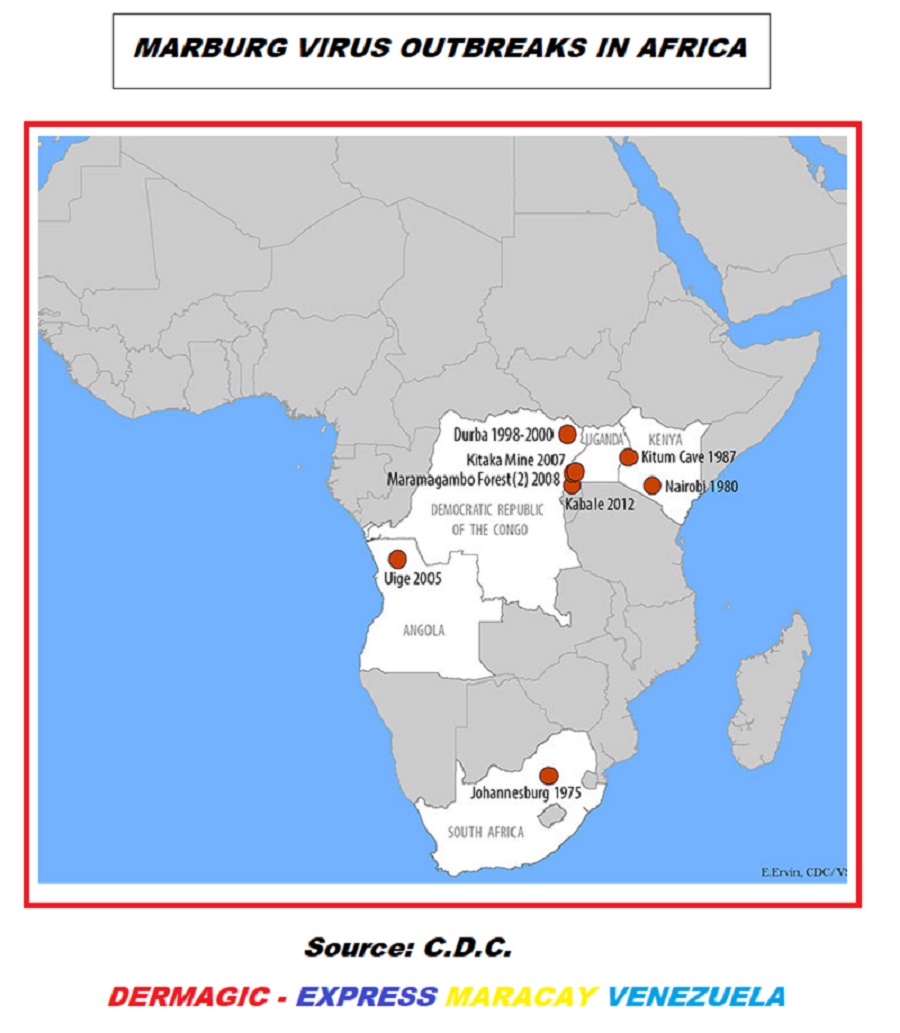
Subsequent outbreaks appeared on the African
continent:
1.)
SOUTH AFRICA: In
Johannesburg 1,975
2.)
KENYA:
Nairobi 1.980, and in the
cave of Kitum 1,987,
3.)
RUSSIA: in
Koltsovo in 1.998-98.
4.)
DEMOCRATIC REPUBLIC OF CONGO: in
Durba and
Watsa, 1.998- 2,000.
5.)
UGANDA:
Kitaka Mine 2.007 and
Forest of Maramagambo 2.008.
6.)
NETHERLANDS AND THE UNITED STATES: 2 cases
imported 2.008.
6.)
ANGOLA: in the
province of Uije 2.004-2.005
7.)
UGANDA: in
Kabale 2.012.
8.)
UGANDA: in 2.014.
The family of the
FILOVIRUS consists of 8 different species:
EBOLA VIRUS, LLOVIU VIRUS, MARBURG VIRUS, RESTON
VIRUS, SUDAN VIRUS, TAI VIRUS, BUNDIBUGYO VIRUS
and RAVN VIRUS, of these RAVN
VIRUS is considered a close relative of
MARBURG VIRUS AND ALSO It causes
HEMORRHAGIC FEVER disease (MVD) by
MARBURG VIRUS.
This virus is considered by the CDC (Centers
for Disease Control and Prevention)
a pathogen CATEGORY A of BIOTERRORISM agent
in whose list are:
TULAREMIA, ANTRAX, SMALLPOX, BOTULINUM TOXIN,
BUBONIC PLAGUE and HEMORRAGIC VIRAL FEVERS
which includes members of the family
FILOVIRIDAE (Marburg and Ebola)
and the family
ARENAVIRIDAE (Lassa virus and Machupo Virus).
The TRANSMISSION of MARBURG VIRUS
initially as I told you was the contact of HUMANS
with tissues of AFRICAN GREEN MONKEYS infected in
1.967; Later it was verified that the
BATS of the African FRUIT (Rousettus
aegyptiacus) IS
CARRIER OF THE MARBURG VIRUS(MARV).
There were outbreaks in Miners in the
KITAKA CAVES in
UGANDA, TOURISTS that
visited the
PHYTON cave
in the same country, and other CAVES
of Africa:
KITUM CAVE in
KENYA. In 2008, a
DUTCH tourist became ill with the MARBURG
VIRUS after visiting the PHYTON cave in
UGANDA; it is believed that the
feces pulverized in the environment were the
mechanism of contagion. Later it was
demonstrated that this BAT is the NATURAL
RESERVOIR of the
MARBUG VIRUS, and also of the
EBOLA VIRUS.
It is also suspected that primates may also be a
source of infection but have not been shown to be
NATURAL RESERVOIRS.
HUMAN-HUMAN transmission was also
demonstrated through direct contact with
blood or other body fluids such as
SALIVA, SWEAT, FECES, URINE, TEARS, AND
BREAST MILK from infected patients. Even
the virus has been found in
TEARS, SEMEN, and HEPATIC BIOPSY weeks
to months after infection, this being important for
the monitoring of convalescent patients.
CONTACT with infected patients and
HANDLING of cadavers is also a source of
infection and therefore there are rigid protocols
for manipulation of patients.
SYMPTOMS OF MARBURG VIRUS DISEASE (MVD):
==========================================
The DISEASE is called
HEMORRHAGIC FEVER (HF) BY MARBURG VIRUS (MARV),
but it should be noted that the RAVN VIRUS in
addition to the MARBURG VIRUS is also causal agent
of the same.
The
INCUBATION Period: has an average of
5-9 days but can reach 21 days. Then a
phase of
GENERALIZATION is presented:
(day 1-5) with HEADACHES, CHILLS,
NAUSEAS, VOMITING, FATIGUE, DIARRHEA, CUTANEOUS
ERUPTION, ABDOMINAL PAIN, MALAISE. Then there is
an
EARLY ORGANIC phase,
(day 5-13) which includes DYSPNEA,
EDEMA CONJUNCTIVAL INJECTION, EXANTHEMA, CNS
SYMPTOMS: ENCEPHALITIS, CONFUSION, DELIRIUM, APATHY,
AGRESSION, then there are
HEMORRHAGIC symptoms that produce
hypovolemia, FECES WITH BLOOD, ECCHYMOSES,
HEMATEMSESIS, HEMORRHAGES OF MUCOSAS AND VISCERAL,
LOSS OF BLOOD IN THE VENIPUNCTURE SITES, in this
phase the patient takes two routes: recovery or
death.
The
POPULAR belief is that the patient
dies for HEMORRHAGES and this is not true.
The patient goes to the
LATE ORGANIC stage
(day 13 - 21) where he enters
CONVALESCENCE or continuous deteriorating
state presenting PERSISTENT FEVER,
CONVULSIONS, COMA, DISSEMINATED INTRAVASCULAR
COAGULATION (CID), METABOLIC DISTURB,
SHOCK AND DEATH that usually occurs
between the day
8-16.
The MORTALITY for MARBUG VIRUS oscillates
between
80-90%, for some scientists it is the
second most LETAL virus for HUMANITY after EBOLA
VIRUS.
TREATMENT
==========
There is no specific treatment against
Marburg Virus disease or
VACCINES available. Treatment is based on
maintaining electrolytes, fluids, blood replacement,
coagulation factors, blood pressure ... and pray
a lot for the patient to save his life.! so that..
The famous character
BATMAN lives in a
CAVE WHERE ALSO LIVES BATS hence his
name, and goes out at night to fight against the BAD
PEOPLE of GOTIC city (GOTHAM), hopefully never
get infected with
MARBURG VIRUS, as it would be the end of the
movie...
MORAL or LESSON: In the case of MARBURG VIRUS,
the man once again invaded the natural habitat of
the NATURAL RESERVOIR, the
BATS on the CAVES, in search of precious
metals ,,, and encountered
disease
and DEATH.. And you, if you are a
tourist and you are going to VISIT A CAVE
look at the ROOF and avoid the BATS. !
Greetings to all.
Dr. José Lapenta
EDITORIAL ESPAÑOL
=================
Hola amigos de la red DERMAGIC EXPRESS te trae hoy
un tema bien interesante, se trata de EL VIRUS
MARBURG(MARV), LA NOCHE DE LOS MURCIELAGOS,
virus hoy día considerado
UNO DE LOS VIRUS MAS LETALES para la HUMANIDAD,
luego del
VIRUS EBOLA, aunque ambos pertenecen a la
misma familia de FILOVIRUS.
Pero quizá lo más
SORPRENDENTE que encontré en esta
revisión es que este virus en los años de la GUERRA
FRIA en la UNION SOVIETICA fue uno de los tantos
agentes biológicos que trato de ser utilizado como
ARMA BIOLOGICA,
para ser colocado en las
ojivas nucleares de los misiles Rusos.
Hecho denunciado por el científico Ruso Ken
Alibek, quien trabajo en uno de los laboratorios
en los años 1.983-1.988 para tales efectos y
que deserto a los Estados Unidos de Norteamérica
posteriormente y revelo estos secretos de Estado.
Otro detalle a considerar si te leíste las
revisiones previas, es que el hábitat donde
vivimos los humanos
NO ES TAN INOFENSIVO, estamos rodeados
por
GARRAPATAS (Enfermedad de
Lyme, Powassan, Heartland, Fiebre de Crimea-Congo
y otras)
INSECTOS
(Zika, Dengue, Chikungunya, Fiebre del Nilo,
Encefalitis Japonesa, jamestown canyon virus
y otros),
RATAS, RATONES (Hantavirus,
Peste bubonica, Leptospirosis, Salmonella y
otras) TRANSMISORES de virus que son
altamente MORTALES o te inoculan una
enfermedad que quizá te deje secuelas de por vida. Y
hoy te traigo además de estos,
LOS MURCIELAGOS, inofensivos MAMIFEROS
VOLADORES que habitan en cuevas, y de actividad
nocturna, RESERVORIOS NATURALES DEL VIRUS DE
MARBURG y EBOLA considerados hoy día los 2
virus
MAS LETALES para la humanidad con una
mortalidad superior al 80%. Luego del
contagio.
EL VIRUS MARBURG (MARV) pertenece al grupo
ARN VIRUS, orden:
MONONEGAVIRALES, familia:
FILOVIRIDAE, genus:
MARBURGVIRUS, especie:
MARGBURG MARBURGVIRUS.
El VIRUS de MARBURG apareció por primera vez en
1.967 en UN LABORATORIO en las localidades de
MARBURG Y
FRANKFURT en
Alemania y en
Belgrado, Yugoslavia (hoy Serbia), cuando
trabajadores se infectaron por un patógeno
desconocido que provoco manifestaciones
HEMORRAGICAS y alta letalidad. El agente causal
fue aislado e identificado por científicos de la
Universidad de MARBURG, Alemania de allí
su nombre. Los portadores del virus fueron
MONOS VERDES AFRICANOS (Chlorocebus aethiops)
infectados que habían sido importados para ser
utilizados en la fabricación de vacunas en el
laboratorio alemán HOECHST AG, hoy día SANOFI-AVENTIS.
Posteriormente aparecieron brotes en el
continente Africano:
1.)
SUR AFRICA: En
Johannesburgo 1.975
2.)
KENYA:
Nairobi 1.980, y en la
cueva de Kitum 1.987,
3.)
RUSIA: en
Kosovo en 1.998-1.990.
4.)
REPBUBLICA DEMOCRATICA DEL CONGO: en
Durba y
Watsa, 1.998-2.000.
5.)
UGANDA:
Mina de Kitaka 2.007 y
Bosque de Maramagambo 2.008.
6.)
HOLANDA Y ESTADOS UNIDOS: 2 casos importados
2.008.
6.)
ANGOLA: en la
provincia de Uije 2.004-2.005
7.)
UGANDA: en
Kabale 2.012.
8.)
UGANDA: en 2.014.
La familia de los
FILOVIRUS está compuesta por 8 distintas
especies:
EBOLA VIRUS, LLOVIU VIRUS, MARBURG VIRUS, RESTON
VIRUS, SUDAN VIRUS, TAI VIRUS, BUNDIBUGYO VIRUS
y RAVN VIRUS,
de estos
RAVN VIRUS es considerado un
pariente cercano del MARBURG VIRUS Y TAMBIEN
OCASIONA la enfermedad
FIEBRE HEMORRAGICA POR MARBURG VIRUS.
Este virus está considerado por el CDC
(Centros de Control y Prevención de Enfermedades)
un patógeno CATEGORIA A de agente de
BIOTERRORISMO en cuya lista están:
TULAREMIA, ANTRAX, VIRUELA, TOXINA BOYULINICA,
PESTE BUBONICA y FIEBRES VIRALES HEMORRAGICAS
que incluye los miembros de la familia
FILOVIRIDAE (Marburg y Ebola) y de la
familia
ARENAVIRIDAE (Lassa virus y Machupo Virus).
La TRANSMISION de MARBURG VIRUS
inicialmente como les comente fue el contacto de
HUMANOS con tejidos de MONOS VERDES AFRICANOS
infectados en 1.967; Posteriormente se comprobó
que el
MURCIELAGO de la FRUTA africano (Rousettus
aegyptiacus) ES PORTADOR DEL
VIRUS DE MARBURG(MARV).
Se presentaron brotes en Mineros en las
CUEVAS de KITAKA en
UGANDA, TURISTAS que visitaron
la cueva PHYTON en el mismo país, y otras
CUEVAS de África:
CUEVA KITUM en
KENYA. En 2.008 un
turista HOLANDES se enfermo con el
VIRUS DE MARBURG luego de visitar la cueva PHYTON en
UGANDA, se cree que
las heces pulverizadas en el medio ambiente
fueron el mecanismo de contagio. Posteriormente
se demostró que este MURCIELAGO es el RESERVORIO
NATURAL del
MARBUG VIRUS, y también del
EBOLA VIRUS.
Se sospecha tambien que los primates tambien pueden
ser fuente de infeccion pero no se ha demostrado que
estos sean RESERVORIOS NATURALES.
También se comprobó la transmisión
HUMANO-HUMANO a través de del contacto
directo con sangre u otros fluidos corporales como
SALIVA, SUDOR, HECES, ORINA, LAGRIMAS
y LECHE MATERNA de pacientes
infectados. Incluso el virus se ha encontrado en
LAGRIMAS, SEMEN, y
BIOPSIA HEPATICA semanas a meses
después de la infección, siendo esto importante para
el monitoreo de pacientes convalecientes. El
CONTACTO con los pacientes infectados
y la
MANIPULACION de cadáveres también es
fuente de infección y por ello existen protocolos
rígidos para la manipulación de los pacientes.
SINTOMAS DE LA ENFERMEDAD MARBURG VIRUS (MVD):
================================================
La ENFERMEDAD es denominada
FIEBRE HEMORRAGICA (HF) POR MARBURG VIRUS (MARV),
pero hay que resaltar que el VIRUS RAVN además del
MARBURG VIRUS es también agente causal de la misma.
El Periodo de
INCUBACION: tiene un promedio de
5-9 días pero puede alcanzar los 21 días.
Luego se presenta una fase de
GENERALIZACION:
(día 1-5) con CEFALEAS, ESCALOSFRIOS,
NAUSEAS, VOMITOS, DIARREA, ERUPCION CUTANEA, DOLOR
ABDOMINAL, MALESTAR GENERAL. Luego se presenta
una
fase TEMPRANA ORGANICA,
(día 5-13) que incluyen DISNEA,
EDEMA CONJUNTIVITIS, EXANTEMA, SINTOMAS DEL SNC:
ENCEFALITIS, CONFUSION, DELIRIO, APATIA, AGRESION,
luego se presentan los síntomas
HEMORRAGICOS que producen hipovolemia,
HECES CON SANFRE, PETEQUIAS, EQUIMOSIS,
HEMATEMSESIS, HEMORRAGIAS DE MUCOSAS Y VISCERAL,
PERDIDA DE SANGRE EN LOS SITIOS DE VENOPUNCION,
en esta fase el paciente toma dos vías: la
recuperación o la muerte.
La creencia POPULAR es que el paciente
muere por las HEMORRAGIAS y no es cierto
esto. El paciente pasa a la
fase TARDIA ORGANICA
(día 13 -21) donde entra en estado de
CONVALESCENCIA o continua deteriorándose
presentando PIEBRE PERSISTENTE, CONVULSIONES,
COMA, COAGULACION INTRAVASCULAR DISEMINADA (CID),
DISTURBIOS METABOLICOS,
SHOCK Y MUERTE
que usualmente se presenta entre el día
8-16.
La MORTALIDAD por el MARBUG VIRUS oscila
entre
80-90%, para algunos científicos es el
segundo virus más LETAL para la HUMANIDAD luego del
EBOLA VIRUS.
TRATAMIENTO:
=============
No hay tratamiento especifico contra la
enfermedad por Marburg Virus
ni VACUNAS disponibles. El tratamiento se
basa en mantener los electrolitos,fluidos,
reemplazar la sangre, factores de la coagulacion,
presion Sanguinea... y rezar mucho para que el
paciente logre salvar su vida. ! De modo que..:
El famoso personaje
HOMBRE-MURCIELAGO (BATMAN) vive en una
CUEVA donde tambien viven LOS MURCIELAGOS,
de allí su nombre, y sale de noche a luchar contra
los BANDIDOS de ciudad GOTICA (GOTHAM), esperemos
que nunca se infecte con
MARBURG VIRUS, pues sería el fin de la
película...
MORALEJA: en el caso de MARBURG VIRUS, el
hombre una vez mas invadio el habitat natural del
RESERVORIO,
LOS MURCIELAGOS en las CUEVAS, en busca
de metales preciosos,,, y se encontro con la
enfermedad y la MUERTE... Y tú, si eres
turista y vas a VISITAR UNA CUEVA
mira para el techo y evita los
MURCIELAGOS!
Saludos a todos.
Dr. José Lapenta
=======================================================================
REFERENCIAS BIBLIOGRAFICAS/
BIBLIOGRAPHICAL REFERENCES
=======================================================================
1.) Filovirus Research: How it Began.
2.) Forty-five years of Marburg virus research.
3.) Marburg haemorrhagic fever in returning
travellers: an overview aimed at clinicians.
4.) Imported case of Marburg hemorrhagic fever -
Colorado, 2008.
5.) Seasonal pulses of Marburg virus circulation
in juvenile Rousettus aegyptiacus bats coincide
with periods of increased risk of human
infection.
6.) Repeated outbreaks of viral hemorrhagic
fevers in Uganda.
7.) Oral shedding of Marburg virus in
experimentally infected Egyptian fruit bats
(Rousettus aegyptiacus).
8.) Mapping the zoonotic niche of Marburg virus
disease in Africa.
9.) Is Marburg virus enzootic in Gabon?
10.) Marburg virus infection detected in a
common African bat.
11.) Studies of reservoir hosts for Marburg
virus.
12.) Marburgvirus Resurgence in Kitaka Mine Bat
Population after Extermination Attempts, Uganda
13.) Marburgvirus genomics and association with
a large hemorrhagic fever outbreak in Angola.
14.) Isolation of genetically diverse Marburg
viruses from Egyptian fruit bats.
15.) Filoviruses and bats.
16.) Outbreak of Marburg hemorrhagic fever among
miners in Kamwenge and Ibanda Districts, Uganda,
2007.
17.) Guide to the Correct Use of Filoviral
Nomenclature.
18.) Biohazard
The Chilling True Story of the Largest Covert
Biological Weapons Program in the World -- Told
from Inside by the Man Who Ran It
19.) Ebola and Marburg virus vaccines.
.===============================================================
===============================================================
1.) Filovirus Research: How it Began.
===============================================================
Curr Top Microbiol Immunol. 2017 Aug 2. doi:
10.1007/82_2017_8. [Epub ahead of print]
Slenczka W1.
Author information
1
Institute of Virology, Philipps University of
Marburg, Am Weinberg 19, 35037, Marburg,
Germany. [email protected].
Abstract
The first reported filovirus outbreak occurred
in August 1967, when laboratory workers in
Marburg and Frankfurt, Germany, and Belgrade,
Yugoslavia (now Serbia) became infected with an
unknown highly pathogenic agent. The disease was
characterized by high fever, malaise, rash,
hemorrhagic and tetanic manifestations, and high
lethality, amounting to 25%. The disease was
introduced to Europe by grivets (Chlorocebus
aethiops), which were used for biomedical
research and vaccine production. The causative
agent, Marburg virus, was isolated and
identified by scientists of the University of
Marburg, Germany in cooperation with specialists
for viral electron microscopy at the Bernhard
Nocht Institute in Hamburg, Germany. In this
chapter, Dr. Slenczka, who was involved in the
first isolation of Marburg virus in 1967,
describes the desperate hunt of the causative
agent of this first filovirus disease outbreak
in the center of Europe, its successful
isolation, the likely route of transmission from
a monkey trading station to vaccine production
facilities in Germany and Yugoslavia, and the
consequences of this outbreak, including a
shortage in the production of poliomyelitis
vaccine In addition, this chapter provides
insight into some of the peculiarities of
filovirus infection, such as sexual virus
transmission several months after recovery and
the role of Ca2+-loss in Marburg virus
pathogenesis, which were already observed during
this first well-documented Marburg virus disease
outbreak.
=========================================================================
2.) Forty-five years of Marburg virus research.
========================================================================
Viruses. 2012 Oct 1;4(10):1878-927. doi:
10.3390/v4101878.
Brauburger K1, Hume AJ, Mühlberger E, Olejnik J.
Author information
1
Department of Microbiology, School of Medicine
and National Emerging Infectious Diseases
Laboratories Institute, Boston University,
Boston, MA 02118, USA. [email protected]
Abstract
In 1967, the first reported filovirus
hemorrhagic fever outbreak took place in Germany
and the former Yugoslavia. The causative agent
that was identified during this outbreak,
Marburg virus, is one of the most deadly human
pathogens. This article provides a comprehensive
overview of our current knowledge about Marburg
virus disease ranging from ecology to
pathogenesis and molecular biology.
=========================================================================
3.) Marburg haemorrhagic fever in returning
travellers: an overview aimed at clinicians.
========================================================================
Clin Microbiol Infect. 2015 Jun 22. pii:
S1198-743X(15)00538-8. doi:
10.1111/1469-0691.12673. [Epub ahead of print]
Bauer MP1, Timen A2, Vossen AC3, van Dissel JT4.
Author information
1
Department of Infectious Diseases, Leiden
University Medical Centre, Leiden, The
Netherlands.
2
National Institute for Public Health and the
Environment, Bilthoven, The Netherlands.
3
Department Medical Microbiology, Leiden
University Medical Centre, Leiden, The
Netherlands.
4
Department of Infectious Diseases, Leiden
University Medical Centre, Leiden, The
Netherlands. Electronic address:
[email protected].
Abstract
Marburg virus haemorrhagic fever (MARV HF) is a
dramatic disease that can occur in a traveller
returning from an area where the virus is
endemic. In this article, we provide an overview
of MARV HF as an imported infection with an
emphasis on clinical aspects. Although late
features such as rash, signs of haemorrhagic
diathesis and liver necrosis may point to the
diagnosis, the initial clinical picture is
non-specific. If in this early phase the
patient's epidemiological exposure history is
compatible with MARV HF, the patient should be
isolated and managed according to viral
haemorrhagic fever protocol and RT-PCR should be
performed on the patient's blood as soon as
possible to rule out MARV HF (or other possible
viral haemorrhagic fevers). In severe cases,
direct electron microscopy of blood in
specialized centres (e.g. Bernhard-Nocht
Institute in Hamburg, Germany) may be considered
if the result of the RT-PCR is not readily
available. Adequate diagnostics and empirical
treatment for other acute life-threatening
illnesses should not be withheld while test
results are awaited, but all management and
diagnostics should be weighed against the risks
of nosocomial transmission.
=========================================================================
4.) Imported case of Marburg hemorrhagic fever -
Colorado, 2008.
========================================================================
Centers for Disease Control and Prevention
(CDC).
Abstract
Marburg hemorrhagic fever (MHF) is a rare, viral
hemorrhagic fever (VHF); the causative agent is
an RNA virus in the family Filoviridae, and
growing evidence demonstrates that fruit bats
are the natural reservoir of Marburg virus
(MARV). On January 9, 2008, an infectious
disease physician notified the Colorado
Department of Public Health and Environment
(CDPHE) of a case of unexplained febrile illness
requiring hospitalization in a woman who had
returned from travel in Uganda. Testing of early
convalescent serum demonstrated no evidence of
infection with agents that cause tropical
febrile illnesses, including VHF. Six months
later, in July 2008, the patient requested
repeat testing after she learned of the death
from MHF of a Dutch tourist who had visited the
same bat-roosting cave as the patient, the
Python Cave in Queen Elizabeth National Park,
Uganda. The convalescent serologic testing
revealed evidence of prior infection with MARV,
and MARV RNA was detected in the archived early
convalescent serum. A public health
investigation did not identify illness
consistent with secondary MHF transmission among
her contacts, and no serologic evidence of
infection was detected among the six tested of
her eight tour companions. The patient might
have acquired MARV infection through exposure to
bat secretions or excretions while visiting the
Python Cave. Travelers should be aware of the
risk for acquiring MHF in caves or mines
inhabited by bats in endemic areas in
sub-Saharan Africa. Health-care providers should
consider VHF among travelers returning from
endemic areas who experience unexplained febrile
illness.
=========================================================================
5.) Seasonal pulses of Marburg virus circulation
in juvenile Rousettus aegyptiacus bats coincide
with periods of increased risk of human
infection.
=========================================================================
PLoS Pathog. 2012;8(10):e1002877. doi:
10.1371/journal.ppat.1002877. Epub 2012 Oct 4.
Amman BR1, Carroll SA, Reed ZD, Sealy TK,
Balinandi S, Swanepoel R, Kemp A, Erickson BR,
Comer JA, Campbell S, Cannon DL, Khristova ML,
Atimnedi P, Paddock CD, Crockett RJ, Flietstra
TD, Warfield KL, Unfer R, Katongole-Mbidde E,
Downing R, Tappero JW, Zaki SR, Rollin PE,
Ksiazek TG, Nichol ST, Towner JS.
Author information
1
Viral Special Pathogens Branch, Centers for
Disease Control and Prevention, Atlanta,
Georgia, United States of America.
Abstract
Marburg virus (family Filoviridae) causes
sporadic outbreaks of severe hemorrhagic disease
in sub-Saharan Africa. Bats have been implicated
as likely natural reservoir hosts based most
recently on an investigation of cases among
miners infected in 2007 at the Kitaka mine,
Uganda, which contained a large population of
Marburg virus-infected Rousettus aegyptiacus
fruit bats. Described here is an ecologic
investigation of Python Cave, Uganda, where an
American and a Dutch tourist acquired Marburg
virus infection in December 2007 and July 2008.
More than 40,000 R. aegyptiacus were found in
the cave and were the sole bat species present.
Between August 2008 and November 2009, 1,622
bats were captured and tested for Marburg virus.
Q-RT-PCR analysis of bat liver/spleen tissues
indicated ~2.5% of the bats were actively
infected, seven of which yielded Marburg virus
isolates. Moreover, Q-RT-PCR-positive lung,
kidney, colon and reproductive tissues were
found, consistent with potential for oral,
urine, fecal or sexual transmission. The
combined data for R. aegyptiacus tested from
Python Cave and Kitaka mine indicate low level
horizontal transmission throughout the year.
However, Q-RT-PCR data show distinct pulses of
virus infection in older juvenile bats (~six
months of age) that temporarily coincide with
the peak twice-yearly birthing seasons.
Retrospective analysis of historical human
infections suspected to have been the result of
discrete spillover events directly from nature
found 83% (54/65) events occurred during these
seasonal pulses in virus circulation, perhaps
demonstrating periods of increased risk of human
infection. The discovery of two tags at Python
Cave from bats marked at Kitaka mine, together
with the close genetic linkages evident between
viruses detected in geographically distant
locations, are consistent with R. aegyptiacus
bats existing as a large meta-population with
associated virus circulation over broad
geographic ranges. These findings provide a
basis for developing Marburg hemorrhagic fever
risk reduction strategies.
========================================================================
6.) Repeated outbreaks of viral hemorrhagic
fevers in Uganda.
========================================================================
Mbonye A1, Wamala J, Winyi-Kaboyo, Tugumizemo V,
Aceng J, Makumbi I.
Author information
1
Ministry of Health Head Quarters, P.O Box 7272
Kampala, Uganda. [email protected]
Abstract
BACKGROUND:
Since the year 2000, Uganda has experienced
repeated outbreaks of viral hemorrhagic fevers
(VHF). Ebola VHF outbreak occurred in the
districts of Gulu in 2000, Bundibugyo, 2007,
Luwero, 2011, Kibaale in July 2012, Luwero in
November 2012. Marburg VHF was earlier reported
in Ibanda in 2007. More recently in 2012, two
outbreaks of Marburg VHF have occurred in Ibanda
and Kabale districts.
OBJECTIVE:
To present the epidemiological picture of the
Marburg VHF recently reported in Ibanda and
Kabale districts and propose research questions
to generate evidence to mitigate future
epidemics.
METHODS:
A case definition for a VHF was developed. A
frequency distribution of symptoms of confirmed
and probable cases was done. Descriptive
analyses of reported cases using simple
percentages, percent distributions and
computation of means was performed.
RESULTS:
The Marburg epidemic was reported in early
September and by November 2012, a cumulative of
14 cases (9 confirmed and 5 probable) including
7 deaths had been registered, giving a case
fatality rate (CFR) of 50%. A total of 202
contacts had been listed; out of which 193 had
completed the 21-day follow-up period. The index
case was a 33-year old male, a teacher at
Nyakatukura Secondary School in Ibanda district.
He travelled to Ibanda from Kabale, his home
district on 31st August 2012, reportedly
healthy. He fell sick on 3rd September 2012 with
complaints of fever, headache, loss of appetite
and general body weakness. Overall, the dominant
symptoms for all cases were fever, vomiting,
loss of appetite, headache, abdominal pain,
fatigue, diarrhea, and the least in occurrence
was bleeding which accounted for 35.5% of all
the cases.
CONCLUSION:
The source of infection for all the five Ebola
Hemorrhagic fever outbreaks in Uganda and the
recent Marburg VHF outbreak in Ibanda and Kabale
is not known. Currently there is suspicion that
there could be an animal reservoir of the Ebola
and Marburg viruses from where occasional
spillage into the human population occurs
resulting in disease outbreaks. This and other
hypotheses require further investigation.
========================================================================
7.) Oral shedding of Marburg virus in
experimentally infected Egyptian fruit bats
(Rousettus aegyptiacus).
========================================================================
J Wildl Dis. 2015 Jan;51(1):113-24. doi:
10.7589/2014-08-198.
Amman BR1, Jones ME, Sealy TK, Uebelhoer LS,
Schuh AJ, Bird BH, Coleman-McCray JD, Martin BE,
Nichol ST, Towner JS.
Author information
1
1 Centers for Disease Control and Prevention,
Viral Special Pathogens Branch, 1600 Clifton Rd.
NE, Atlanta, Georgia 30333, USA.
Abstract
Marburg virus (Marburg marburgvirus; MARV)
causes sporadic outbreaks of Marburg hemorrhagic
fever (MHF) in Africa. The Egyptian fruit bat
(Rousettus aegyptiacus) has been identified as a
natural reservoir based most-recently on the
repeated isolation of MARV directly from bats
caught at two locations in southwestern Uganda
where miners and tourists separately contracted
MHF from 2007-08. Despite learning much about
the ecology of MARV through extensive field
investigations, there remained unanswered
questions such as determining the primary routes
of virus shedding and the severity of disease,
if any, caused by MARV in infected bats. To
answer these questions and others, we
experimentally infected captive-bred R.
aegyptiacus with MARV under high (biosafety
level 4) containment. These experiments have
shown infection profiles consistent with R.
aegyptiacus being a bona fide natural reservoir
host for MARV and demonstrated routes of viral
shedding capable of infecting humans and other
animals.
========================================================================
8.) Mapping the zoonotic niche of Marburg virus
disease in Africa.
=======================================================================
Trans R Soc Trop Med Hyg. 2015
Jun;109(6):366-78. doi: 10.1093/trstmh/trv024.
Epub 2015 Mar 27.
Pigott DM1, Golding N2, Mylne A2, Huang Z2,
Weiss DJ2, Brady OJ2, Kraemer MU2, Hay SI3.
Author information
1
Spatial Ecology & Epidemiology Group, Department
of Zoology, University of Oxford, Oxford, UK
[email protected].
2
Spatial Ecology & Epidemiology Group, Department
of Zoology, University of Oxford, Oxford, UK.
3
Spatial Ecology & Epidemiology Group, Department
of Zoology, University of Oxford, Oxford, UK
Fogarty International Center, National
Institutes of Health, Bethesda, Maryland, USA.
Abstract
BACKGROUND:
Marburg virus disease (MVD) describes a viral
haemorrhagic fever responsible for a number of
outbreaks across eastern and southern Africa. It
is a zoonotic disease, with the Egyptian
rousette (Rousettus aegyptiacus) identified as a
reservoir host. Infection is suspected to result
from contact between this reservoir and human
populations, with occasional secondary
human-to-human transmission.
METHODS:
Index cases of previous human outbreaks were
identified and reports of infection in animals
recorded. These data were modelled within a
species distribution modelling framework in
order to generate a probabilistic surface of
zoonotic transmission potential of MVD across
sub-Saharan Africa.
RESULTS:
Areas suitable for zoonotic transmission of MVD
are predicted in 27 countries inhabited by 105
million people. Regions are suggested for
exploratory surveys to better characterise the
geographical distribution of the disease, as
well as for directing efforts to communicate the
risk of practices enhancing zoonotic contact.
CONCLUSIONS:
These maps can inform future contingency and
preparedness strategies for MVD control,
especially where secondary transmission is a
risk. Coupling this risk map with patient travel
histories could be used to guide the
differential diagnosis of highly transmissible
pathogens, enabling more rapid response to
outbreaks of haemorrhagic fever.
========================================================================
9.) Is Marburg virus enzootic in Gabon?
========================================================================
J Infect Dis. 2011 Nov;204 Suppl 3:S800-3. doi:
10.1093/infdis/jir358.
Maganga GD1, Bourgarel M, Ella GE, Drexler JF,
Gonzalez JP, Drosten C, Leroy EM.
Author information
1
Centre International de Recherches Médicales de
Franceville, Gabon.
Abstract
Marburg virus (MARV) nucleic acid was detected
in Rousettus aegyptiacus bats in 2005 and 2006
in the midwest and southeast of Gabon. In this
study we used MARV-specific real-time
reverse-transcription polymerase chain reaction
(RT-PCR) and MARV-specific nested RT-PCR assay
to screen 1257 bats caught during July 2009,
December 2009, and June 2010 in 3 caves situated
in northern Gabon. Nine specimens tested
positive by the real-time assay, with cycle
threshold values ranging from 35 to 39, of which
only 1 R. aegyptiacus specimen collected in 2009
was positive in the nested VP35 RT-PCR assay.
Together with MARV-positive bats in the south
and west found in 2005 and 2006, confirmation of
phylogenetically closely related MARV-positive
bats 5 years later and in northern Gabon
suggests that MARV is now enzootic in Gabon and
emphasizes the importance of long-term
monitoring of bat populations and human-bat
interfaces.
========================================================================
10.) Marburg virus infection detected in a
common African bat.
========================================================================
PLoS One. 2007 Aug 22;2(8):e764.
Towner JS1, Pourrut X, Albariño CG, Nkogue CN,
Bird BH, Grard G, Ksiazek TG, Gonzalez JP,
Nichol ST, Leroy EM.
Author information
1
Centers for Disease Control and Prevention,
Special Pathogens Branch, Atlanta, Georgia,
United States of America.
Abstract
Marburg and Ebola viruses can cause large
hemorrhagic fever (HF) outbreaks with high case
fatality (80-90%) in human and great apes.
Identification of the natural reservoir of these
viruses is one of the most important topics in
this field and a fundamental key to
understanding their natural history. Despite the
discovery of this virus family almost 40 years
ago, the search for the natural reservoir of
these lethal pathogens remains an enigma despite
numerous ecological studies. Here, we report the
discovery of Marburg virus in a common species
of fruit bat (Rousettus aegyptiacus) in Gabon as
shown by finding virus-specific RNA and IgG
antibody in individual bats. These Marburg virus
positive bats represent the first naturally
infected non-primate animals identified.
Furthermore, this is the first report of Marburg
virus being present in this area of Africa, thus
extending the known range of the virus. These
data imply that more areas are at risk for MHF
outbreaks than previously realized and
correspond well with a recently published report
in which three species of fruit bats were
demonstrated to be likely reservoirs for Ebola
virus.
========================================================================
11.) Studies of reservoir hosts for Marburg
virus.
========================================================================
Emerg Infect Dis. 2007 Dec;13(12):1847-51. doi:
10.3201/eid1312.071115.
Swanepoel R1, Smit SB, Rollin PE, Formenty P,
Leman PA, Kemp A, Burt FJ, Grobbelaar AA, Croft
J, Bausch DG, Zeller H, Leirs H, Braack LE,
Libande ML, Zaki S, Nichol ST, Ksiazek TG,
Paweska JT; International Scientific and
Technical Committee for Marburg Hemorrhagic
Fever Control in the Democratic Republic of
Congo.
Author information
1
National Institute for Communicable Diseases,
Sandringham, Republic of South Africa.
[email protected]
Abstract
To determine reservoir hosts for Marburg virus
(MARV), we examined the fauna of a mine in
northeastern Democratic Republic of the Congo.
The mine was associated with a protracted
outbreak of Marburg hemorrhagic fever during
1998-2000. We found MARV nucleic acid in 12
bats, comprising 3.0%-3.6% of 2 species of
insectivorous bat and 1 species of fruit bat. We
found antibody to the virus in the serum of 9.7%
of 1 of the insectivorous species and in 20.5%
of the fruit bat species, but attempts to
isolate virus were unsuccessful
========================================================================
12.) Marburgvirus Resurgence in Kitaka Mine Bat
Population after Extermination Attempts, Uganda
=======================================================================
Emerg Infect Dis. 2014 Oct; 20(10): 1761–1764.
doi: 10.3201/eid2010.140696
Brian R. Amman, Luke Nyakarahuka, Anita K.
McElroy, Kimberly A. Dodd, Tara K. Sealy, Amy J.
Schuh, Trevor R. Shoemaker, Stephen Balinandi,
Patrick Atimnedi, Winyi Kaboyo, Stuart T.
Nichol, and Jonathan S. Townercorresponding
author
To the Editor: Marburg virus (MARV) and Ravn
virus (RAVV), collectively called
marburgviruses, cause Marburg hemorrhagic fever
(MHF) in humans. In July 2007, 4 cases of MHF (1
fatal) occurred in miners at Kitaka Mine in
southern Uganda. Later, MHF occurred in 2
tourists who visited Python Cave, ≈50 km from
Kitaka Mine. One of the tourists was from the
United States (December 2007) and 1 was from the
Netherlands (July 2008); 1 case was fatal
(1,2,3). The cave and the mine each contained
40,000–100,000 Rousettus aegyptiacus bats
(Egyptian fruit bats).
Longitudinal investigations of the outbreaks at
both locations were initiated by the Viral
Special Pathogens Branch of the Centers for
Disease Control and Prevention (CDC, Atlanta,
GA, USA, and Entebbe, Uganda) in collaboration
with the Uganda Wildlife Authority (UWA) and the
Uganda Virus Research Institute (UVRI). During
these studies, genetically diverse MARVs and
RAVVs were isolated directly from bat tissues,
and infection levels of the 2 viruses were found
to increase in juvenile bats on a predictable
bi-annual basis (4,5). However, investigations
at Kitaka Mine were stopped when the miners
exterminated the bat colony by restricting
egress from the cave with papyrus reed barriers
and then entangling the bats in fishing nets
draped over the exits. The trapping continued
for weeks, and the entrances were then sealed
with sticks and plastic. These depopulation
efforts were documented by researchers from
UVRI, the CDC, the National Institute of
Communicable Diseases (Sandringham, South
Africa), and UWA during site visits to Kitaka
Mine (Technical Appendix Figure). In August
2008, thousands of dead bats were found piled in
the forest, and by November 2008, there was no
evidence of bats living in the mine; whether
100% extermination was achieved is unknown. CDC,
UVRI, and UWA recommended against extermination,
believing that any results would be temporary
and that such efforts could exacerbate the
problem if bat exclusion methods were not
complete and permanent (6,7).
In October 2012, the most recent known
marburgvirus outbreak was detected in Ibanda, a
town in southwest Uganda. Ibanda is ≈20 km from
the Kitaka Mine and is the urban center that
serves smaller communities in the Kitaka area.
This MHF outbreak was the largest in Ugandan
history: 15 laboratory-confirmed cases occurred
(8). In November 2012, an ecologic investigation
of the greater Ibanda/Kitaka area was initiated.
The investigation included interviews with local
authorities to locate all known R. aegyptiacus
colonies in the area. Although minor colonies of
small insectivorous bats were found, the only
identifiable colony of R. aegyptiacus bats was
found inside the re-opened Kitaka Mine, albeit
at much reduced size, perhaps 1%–5% of that
found before depopulation efforts.
To determine whether the R. aegyptiacus bats
that had repopulated Kitaka Mine were actively
infected with marburgviruses, we tested 400 bats
by using previously described methods (4,5).
Viral RNA was extracted from ≈100 mg of liver
and spleen tissue by using the MagMAX Total
Nucleic Acid Isolation Kit (Applied Biosystems,
Foster City, CA, USA) according to the
manufacturer’s recommended protocol. The Fisher
exact test was conducted by using IBM SPSS
Statistics, version 19.0 (IBM Corp., Armonk, NY,
USA).
Of the 400 R. aegyptiacus bats collected, 53
(13.3%) were positive for marburgvirus RNA by
quantitative reverse transcription PCR (32/233
[13.7%] adults and 21/167 [12.6%] juveniles;
Technical Appendix Table); marburgvirus was
isolated from tissue samples from 9 of the 400
bats. The overall level of active infection was
significantly higher than that found in Kitaka
Mine during 2007–2008 (5.1%) (5) (Fisher exact
test, p<0.001) and in other studies in Uganda
(Python Cave [2.5%]) and Gabon (4.8%) (4,9). The
reason for the increase is not clear, but it may
be related to the effects of the extermination
and subsequent repopulation. Increases in
disease prevalence in wildlife populations after
culling are not unprecedented (6,7). We
speculate that after the depopulation attempt, a
pool of susceptible bats became established over
time and was subjected to multiple marburgvirus
introductions, as evidenced by the genetic
diversity of viruses isolated from the bats
(Figure). A pool of susceptible bats would have
led to higher levels of active infection within
the colony, thereby increasing the potential for
virus spillover into the human population. A
significant sex and age bias was not detected
with respect to active infection during the
breeding season (Fisher exact test, p>0.5 for
both), and overall, the presence of
virus-specific IgG among the bats was 16.5%, a
finding consistent with that in previous studies
(4,5).
Phylogeny of concatenated marburgvirus
nucleoprotein (NP) and viral protein 35 (VP35)
gene fragments as determined by using the
maximum-likelihood method. Sequences from the NP
(289–372 nt) and VP35 (203–213 nt) genes were
amplified and ...
Phylogenetic analysis of viral RNA genome
fragment sequences in this study showed high
marburgvirus genetic diversity, including the
presence of RAVVs and MARVs. Sequences for
isolates from 3 bats were nearly identical to
those of the MARV isolates obtained from
patients in the 2012 Ibanda outbreak (8),
suggesting that bats from Kitaka Mine were a
likely source of the virus.
========================================================================
13.) Marburgvirus genomics and association with
a large hemorrhagic fever outbreak in Angola.
=======================================================================
J Virol. 2006 Jul;80(13):6497-516.
Towner JS1, Khristova ML, Sealy TK, Vincent MJ,
Erickson BR, Bawiec DA, Hartman AL, Comer JA,
Zaki SR, Ströher U, Gomes da Silva F, del
Castillo F, Rollin PE, Ksiazek TG, Nichol ST.
Author information
1
Special Pathogens Branch, Centers for Disease
Control and Prevention, 1600 Clifton Road,
Mailstop G14, Atlanta, GA 30333, USA.
Abstract
In March 2005, the Centers for Disease Control
and Prevention (CDC) investigated a large
hemorrhagic fever (HF) outbreak in Uige Province
in northern Angola, West Africa. In total, 15
initial specimens were sent to CDC, Atlanta,
Ga., for testing for viruses associated with
viral HFs known to be present in West Africa,
including ebolavirus. Marburgvirus was also
included despite the fact that the origins of
all earlier outbreaks were linked directly to
East Africa. Surprisingly, marburgvirus was
confirmed (12 of 15 specimens) as the cause of
the outbreak. The outbreak likely began in
October 2004 and ended in July 2005, and it
included 252 cases and 227 (90%) fatalities
(report from the Ministry of Health, Republic of
Angola, 2005), making it the largest Marburg HF
outbreak on record. A real-time quantitative
reverse transcription-PCR assay utilized and
adapted during the outbreak proved to be highly
sensitive and sufficiently robust for field use.
Partial marburgvirus RNA sequence analysis
revealed up to 21% nucleotide divergence among
the previously characterized East African
strains, with the most distinct being Ravn from
Kenya (1987). The Angolan strain was less
different ( approximately 7%) from the main
group of East African marburgviruses than one
might expect given the large geographic
separation. To more precisely analyze the virus
genetic differences between outbreaks and among
viruses within the Angola outbreak itself, a
total of 16 complete virus genomes were
determined, including those of the virus
isolates Ravn (Kenya, 1987) and 05DRC, 07DRC,
and 09DRC (Democratic Republic of Congo, 1998)
and the reference Angolan virus isolate
(Ang1379v). In addition, complete genome
sequences were obtained from RNAs extracted from
10 clinical specimens reflecting various stages
of the disease and locations within the Angolan
outbreak. While the marburgviruses exhibit high
overall genetic diversity (up to 22%), only 6.8%
nucleotide difference was found between the West
African Angolan viruses and the majority of East
African viruses, suggesting that the virus
reservoir species in these regions are not
substantially distinct. Remarkably few
nucleotide differences were found among the
Angolan clinical specimens (0 to 0.07%),
consistent with an outbreak scenario in which a
single (or rare) introduction of virus from the
reservoir species into the human population was
followed by person-to-person transmission with
little accumulation of mutations. This is in
contrast to the 1998 to 2000 marburgvirus
outbreak, where evidence of several virus
genetic lineages (with up to 21% divergence) and
multiple virus introductions into the human
population was found.
========================================================================
14.) Isolation of genetically diverse Marburg
viruses from Egyptian fruit bats.
========================================================================
PLoS Pathog. 2009 Jul;5(7):e1000536. doi:
10.1371/journal.ppat.1000536. Epub 2009 Jul 31.
Towner JS1, Amman BR, Sealy TK, Carroll SA,
Comer JA, Kemp A, Swanepoel R, Paddock CD,
Balinandi S, Khristova ML, Formenty PB, Albarino
CG, Miller DM, Reed ZD, Kayiwa JT, Mills JN,
Cannon DL, Greer PW, Byaruhanga E, Farnon EC,
Atimnedi P, Okware S, Katongole-Mbidde E,
Downing R, Tappero JW, Zaki SR, Ksiazek TG,
Nichol ST, Rollin PE.
Author information
1
Special Pathogens Branch, Centers for Disease
Control and Prevention, Atlanta, GA, USA.
Abstract
In July and September 2007, miners working in
Kitaka Cave, Uganda, were diagnosed with Marburg
hemorrhagic fever. The likely source of
infection in the cave was Egyptian fruit bats
(Rousettus aegyptiacus) based on detection of
Marburg virus RNA in 31/611 (5.1%) bats,
virus-specific antibody in bat sera, and
isolation of genetically diverse virus from bat
tissues. The virus isolates were collected nine
months apart, demonstrating long-term virus
circulation. The bat colony was estimated to be
over 100,000 animals using mark and re-capture
methods, predicting the presence of over 5,000
virus-infected bats. The genetically diverse
virus genome sequences from bats and miners
closely matched. These data indicate common
Egyptian fruit bats can represent a major
natural reservoir and source of Marburg virus
with potential for spillover into humans.
========================================================================
15.) Filoviruses and bats.
========================================================================
Microbiol. Aust. 2017 Mar;38(1):12-16. doi:
10.1071/MA17005. Epub 2017 Feb 17.
Schuh AJ1, Amman BR1, Towner JS1.
Author information
1
Viral Special Pathogens Branch, Division of
High-Consequence Pathogens and Pathology,
Centers for Disease Control and Prevention,
Atlanta, GA 30329, USA.
Abstract
While Reston and Lloviu viruses have never been
associated with human disease, the other
filoviruses cause outbreaks of hemorrhagic fever
characterised by person-to-person transmission
and high case fatality ratios. Cumulative
evidence suggests that bats are the most likely
reservoir hosts of the filoviruses. Ecological
investigations following Marburg virus disease
outbreaks associated with entry into caves
inhabited by Rousettus aegyptiacus bats led to
the identification of this bat species as the
natural reservoir host of the marburgviruses.
Experimental infection of R. aegyptiacus with
Marburg virus has provided insight into the
natural history of filovirus infection in bats
that may help guide the search for the reservoir
hosts of the ebolaviruses.
========================================================================
16.) Outbreak of Marburg hemorrhagic fever among
miners in Kamwenge and Ibanda Districts, Uganda,
2007.
=======================================================================
J Infect Dis. 2011 Nov;204 Suppl 3:S796-9. doi:
10.1093/infdis/jir312.
Adjemian J1, Farnon EC, Tschioko F, Wamala JF,
Byaruhanga E, Bwire GS, Kansiime E, Kagirita A,
Ahimbisibwe S, Katunguka F, Jeffs B, Lutwama JJ,
Downing R, Tappero JW, Formenty P, Amman B,
Manning C, Towner J, Nichol ST, Rollin PE.
Author information
1
Laboratory of Clinical Infectious Diseases,
National Institute of Allergy and Infectious
Diseases, National Institutes of Health,
Bethesda, Maryland 20892-2665, USA.
[email protected]
Abstract
Marburg hemorrhagic fever was detected among 4
miners in Ibanda District, Uganda, from June
through September, 2007. Infection was likely
acquired through exposure to bats or bat
secretions in a mine in Kamwenge District,
Uganda, and possibly human-to-human transmission
between some patients. We describe the
epidemiologic investigation and the health
education response.
========================================================================
17.) Guide to the Correct Use of Filoviral
Nomenclature.
========================================================================
Curr Top Microbiol Immunol. 2017 Jun 27. doi:
10.1007/82_2017_7. [Epub ahead of print]
Kuhn JH1.
Author information
1
Integrated Research Facility at Fort Detrick
(IRF-Frederick), Division of Clinical Research
(DCR), National Institute of Allergy and
Infectious Diseases (NIAID) National Institutes
of Health (NIH), B-8200 Research Plaza, Fort
Detrick, Frederick, MD, 21702, USA.
[email protected].
Abstract
The International Committee on Taxonomy of
Viruses (ICTV) currently recognizes three genera
and seven species as part of the mononegaviral
family Filoviridae. Eight distinct filoviruses
(Bundibugyo virus, Ebola virus, Lloviu virus,
Marburg virus, Ravn virus, Reston virus, Sudan
virus, and Taï Forest virus) have been assigned
to these seven species. This chapter briefly
summarizes the status quo of filovirus
classification and focuses on the importance of
differentiating between filoviral species and
filoviruses and the correct use of taxonomic and
vernacular filovirus names and abbreviations in
written and oral discourse
========================================================================
18.) Biohazard
The Chilling True Story of the Largest Covert
Biological Weapons Program in the World -- Told from
Inside by the Man Who Ran It
========================================================================
By KEN ALIBEK with STEPHEN HANDELMAN
Source:http://www.nytimes.com/books/99/06/20/reviews/990620.20taubmat.html
Tularemia is a highly infectious disease that
produces headaches, nausea and high fevers. It can
be lethal if untreated. Tularemia is also hard to
extinguish, making it attractive to anyone trying to
produce biological weapons.
That's just what Ken Alibek was doing for the Soviet
Union in 1983 when he found himself standing in a
puddle of tularemia bacteria that had accidentally
spilled onto the floor of a secret weapons lab.
There was enough tularemia in the small, milky brown
pool to infect everyone in the Soviet Union. Within
hours Alibek was too sick to move. Only megadoses of
tetracycline, hastily obtained from a friend,
prevented the disease from disabling if not killing
him.
That is one of many harrowing moments that Alibek
describes in this absorbing account of the Soviet
Union's demonic effort to make biological weapons.
The program was one of the best-kept Russian secrets
of the cold war, and Alibek was one of its central
architects. He reports that at its high point in the
late 1980's, when Mikhail Gorbachev was the Soviet
leader, the program consumed close to $1 billion a
year and employed more than 60,000 people at dozens
of clandestine sites. Needless to say, it was not an
activity that Gorbachev advertised as he tried to
improve relations with the West.
Though the Russian effort is now believed to be
largely abandoned, biological weapons remain a
threat, perhaps even a greater one today because
they can be made relatively easily and inexpensively
by terrorist groups and leaders like Saddam Hussein.
Alibek, born Kanatjan Alibekov, defected to the
United States in 1992 and changed his name. By then
he had quit the weapons project in disgust, but for
nearly all his career as a Soviet scientist he
excelled at the grim business of cultivating
biological agents and adapting them for use in
missiles and bombs. For many years he was deputy
director of Biopreparat, an ostensibly civilian
agency that was actually involved in advanced
research into biological weapons. Alibek provided
American officials with their first full description
of the Soviet effort when he defected.
In ''Biohazard'' he performs the same service for
readers, with a strong writing assist from Stephen
Handelman, who was a Moscow correspondent for The
Toronto Star. The book works best as a richly
descriptive report on the Soviet program and
Alibek's role in it. It is less successful as a
portrait of Alibek and his transition from germ
warfare acolyte to apostate.
The story is sobering. With no limit to the
resources it was prepared to invest in
unconventional weapons research, the Soviet Union
developed an extensive arsenal of deadly pathogens,
including anthrax, smallpox, plague, brucellosis and
tularemia. Tons of these bacteria and viruses were
churned out at production centers, often in
vaccine-resistant strains that could be effectively
dispersed in liquid, powder or aerosol form. Moscow
even tried to manipulate the AIDS virus so it could
be used as a weapon. The disease's long incubation
period made it unsuitable.
For Alibek and his colleagues, the grotesque work
was just another day at the office. He recalls a
meeting in 1988 at Soviet Army headquarters in
Moscow, where he was instructed to arm long-range
missiles with deadly germs. ''I made a few quick
calculations on my note pad,'' he says. ''At least
400 kilograms of anthrax, prepared in dry form for
use as an aerosol, would be required for 10
warheads.'' Martha Stewart couldn't have put it more
innocuously.
The Kremlin went ahead with such work even though it
had signed the 1972 Biological Weapons Convention,
which banned the development, production and
stockpiling of biological agents for offensive
military purposes. Just a year after signing the
accord, the Soviet Government secretly initiated an
effort to modernize its biological weapons and to
invent new ones. The United States, for its part,
maintained a robust biological warfare program until
1969, when President Richard Nixon renounced the use
of such weapons and restricted research to defensive
measures like immunization.
Alibek was drawn into the Soviet campaign in 1975,
deflected from a conventional career as a military
physician by the allure of highly classified
research, the prospect of rapid advancement and the
mistaken belief that the Soviet Union had no choice
but to keep pace with the United States in germ
warfare technology. He was a Kazakh native eager to
prove himself to his Russian superiors. With a knack
for epidemiology and laboratory research, he was
soon building what he describes matter-of-factly as
''the world's most efficient assembly line for the
mass production of weaponized anthrax.''
Alibek has a fine eye for the cold-blooded customs
of the Soviet state, including coercion and
deception. He never told his supervisors about his
frightening bout with tularemia, fearing it would
cost his job. A few years earlier, while a student
at the Tomsk Medical Institute in Siberia, he had
surmised from medical records that Soviet forces had
used the same disease as a weapon against German
troops outside Stalingrad in 1942. His professor, a
colonel, icily told Alibek, ''You have gone beyond
your assignment,'' and advised him never to speak of
the matter again.
Though Alibek struggles to explain his enthusiasm
for biological weapons work, he seems reluctant to
probe beyond surface emotions. He stops the
narrative periodically for moments of introspection
like this: ''I still shuddered occasionally when I
looked at the bacteria multiplying in our fermenters
and considered that they could end the lives of
millions of people. But the secret culture of our
labs had changed my outlook. My parents would not
have recognized the man I had become.'' Unhappily,
these tantalizing passages are but brief
digressions, leaving one to puzzle over just why
Alibek turned against the system.
The Russian program was theoretically dismantled in
recent years at the order of President Boris N.
Yeltsin, but Ken Alibek makes clear there may still
be active remnants. Given the unblinking support he
and thousands of others gave to the effort, that
would not be surprising.
===========================================================================
19.) Ebola and Marburg virus vaccines.
===========================================================================
Virus Genes. 2017 Aug;53(4):501-515. doi:
10.1007/s11262-017-1455-x. Epub 2017 Apr 26.
Reynolds P1, Marzi A2.
Author information
1
Laboratory of Virology, Division of Intramural
Research, National Institute of Allergy and
Infectious Diseases, National Institutes of Health,
Hamilton, MT, USA.
2
Laboratory of Virology, Division of Intramural
Research, National Institute of Allergy and
Infectious Diseases, National Institutes of Health,
Hamilton, MT, USA. [email protected].
Abstract
The filoviruses, Ebola virus (EBOV), and Marburg
virus (MARV), are among the most pathogenic viruses
known to man and the causative agents of viral
hemorrhagic fever outbreaks in Africa with case
fatality rates of up to 90%. Nearly 30,000
infections were observed in the latest EBOV epidemic
in West Africa; previous outbreaks were much smaller,
typically only affecting less than a few hundred
people. Compared to other diseases such as AIDS or
Malaria with millions of cases annually, filovirus
hemorrhagic fever (FHF) is one of the neglected
infectious diseases. There are no licensed vaccines
or therapeutics available to treat EBOV and MARV
infections; therefore, these pathogens can only be
handled in maximum containment laboratories and are
classified as select agents. Under these limitations,
a very few laboratories worldwide conducted basic
research and countermeasure development for EBOV and
MARV since their respective discoveries in 1967 (MARV)
and 1976 (EBOV). In this review, we discuss several
vaccine platforms against EBOV and MARV, which have
been assessed for their protective efficacy in
animal models of FHF. The focus is on the most
promising approaches, which were accelerated in
clinical development (phase I-III trials) during the
EBOV epidemic in West Africa.
=======================================================================
===================================================================
DATA-MEDICOS/DERMAGIC-EXPRESS No 19-(204) 08/10/2.017 DR. JOSE LAPENTA R.
===================================================================
Produced
by Dr. Jose Lapenta R. Dermatologist 2.017
Maracay Estado Aragua Venezuela 2.017
Telf: 0416-6401045- 02432327287-02432328571
loading...
|