| Vaccines, the risk that
represents today. !!
Vacunas, el riesgo que reprensentan hoy
dia. !!
DATA-MEDICOS
DERMAGIC/EXPRESS 19-(203)
22 Octubre 2.017 22 October 2.017
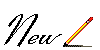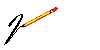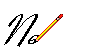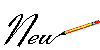
EDITORIAL ENGLISH
=================
Welcome once again all the friends, doctors,
dermatologists, people around the world, readers of
DERMAGIC EXPRESS, today once again talking about the
controversial issue of VACCINES, under the
name NO-VAXX, ALWAYS SAY NEVER AGAIN.
To begin I will say once again that this is not the
first time and perhaps it will not be the last, that
I speak on this subject that today has become one of
the most discussed in the world community, a subject
that has REVIVED by the
ADVERSE EVENTS and
DEATHS caused by VACCINES AGAINST HPV,
GARDASIL and
CERVARIX, cases of
AUTISM AND death caused by some VACCINES,
and to "spill the drop of the glass",
THE NEXT BIRTH OF THE "NEW" VACCINE VL15
of the pharmaceutical Valneva against the LYME
DISEASE.
Firstly, the population must be divided into
three groups: 1.) Those who
do not know or are unaware of the problem of
VACCINES or do not care about them. 2.)
The "VAXERS" those people who agree with the
VACCINES. And 3.) The so-called
ANTI-VAXERS, those people who for one
reason or another
oppose VACCINATIONS, individual and massive,
most of them because a relative or child
suffered ADVERSE EFFECTS or DEATH by some VACCINE,
or personally I was DISABLED or get sick after
some vaccination.
To give you TOTAL credibility to this post I will
start by placing you the
ADVERSE EFFECTS AND
DEATHS REPORTED BY THE ADVERSE EVENTS
NOTIFICATION SYSTEM (VAERS) In the United States
on the
MEASLES VACCINE, RUBELLA AND MUMPS, the well-known
MMR.
MMR VACCINE (MEASLES, RUBELLA and MUMPS):
==========================================
There were reported
75,000 adverse effects including:
IN GENERAL: vomiting, diarrhea, anaphylaxis,
earache, nervous deafness, diabetes, arthritis,
myalgia, encephalitis, febrile seizures, pneumonia
and death; IN DETAIL:
1.)
78 deaths
2.)
85 cases of deafness
3.)
48 cases of decreased eye contact
4.)
92 cases of developmental delay
5.)
855 reported cases of autism
6.)
116 cases of intellectual disability
7.)
401 reports of speech disorders
8.)
276 reports of loss of conscience
9.)
143 cases of encephalitis
10.)
74 cases of meningitis
11.)
111 cases of Guillain-Barré syndrome.
12.)
692 cases of gait disturbance (unable to walk
normally)
13.)
748 cases of hypokinesia (partial or total
loss of muscle movement)
14.)
653 reports of hypotonia (poor muscle tone)
15.)
4874 reports of seizures, including febrile
convulsions and TONIC clonic seizures
16.)
1576 CASES OF CELLULITIS (POTENTIALLY SERIOUS
SKIN INFECTION).
And finally, in some cases, the vaccine has caused
the
SAME diseases it is supposed to
prevent, with the following data reported to VAERS:
17.)
147 cases of measles.
18.)
384 cases of mumps.
19.)
29 cases of rubella.
These numbers are not EXACT because the report is
from 1 to 10% of reactions, therefore they may be
higher.
JAPAN BAN MMR VACCINE (MEASLES, RUBELLAS AND MUMPS):
======================================================
About this
MMR VACCINE maybe you did not know that THE
JAPANESE GOVERNMENT
banned it in the year 1.993
(24 years ago) after 1,800,000 children
were vaccinated with 2 MMR types and a high
percentage presented signs of non-viral meningitis
and other ADVERSE REACTIONS, among which
3 CHILDREN DEAD, AND
8 WITH PERMANENT DISABILITIES: DEAF,
BLINDNESS AND LOSS OF THE CONTROL OF THE
EXTREMITIES. I also quote:
".... Of the 3,969 medical compensation claims
relating to vaccines in the last 30 years, a quarter
had been made by those badly affected by the
combined measles, mumps and rubella vaccine, they
say..."
In 1.999 JAPAN considered using the VACCINE again
but preferred to keep the PROHIBITION and to
use the vaccines against MEASLES, MUMPS and
RUBELLA
individually. The cost is higher, but
it's worth it. SIDE AND DEATH effects are minor.
GARDASIL AND CERVARIX (VACCINES AGAINST HPV):
=============================================
Of the vaccine GARSASIL and CERVARIX and its adverse
effects I have already talked to you enough and the
links you have here:
1.)
STOP THE GARDASIL AND CERVARIX VACCINES AGINST
THE HPV, HUMAN PAPILLOMA VIRUS.
2.)
GARDASIL HPV VACCINE & MERCK LAWSUIT IN COLOMBIA,
POST GARDASIL AND CERVARIX HPV VACCINE SYNDROME.
JAPAN BANNED GARDASIL AND CERVARIX VACCINES (HPV) IN
2.013.
=========================================================
But what you perhaps did not know about the
"VAXERS" side is that JAPAN a world
power PROHIBITED,
"BANNED" the
GARDASIL VACCINE in the year 2.013
due to the report of approximately
2.000 SIDE EFFECTS, hundreds of which were
SERIOUS .
The Japanese reported at that time:
CONVULSIONS, CEREBRAL DAMAGE, BLINDNESS, PARALYSIS,
SPEECH PROBLEMS, PANCREATITIS, LOSS OF SHORT TERM
SPEECH, DEATH, AND OTHERS. And I quote
again:
"... Since the government began offering girls HPV
shots, 1,968 adverse events were reported,
including
358 that were evaluated as serious by a
JMLHW committee. Parents began calling the country’s
health minister and furnishing videos in which girls
who had received the HPV vaccine suffered from
walking disturbances, body tics and seizures.
In other cases many
girls injected with the vaccine fell to the floor,
injuring their head or face and some fracturing
their jaw or teeth,”
And still this "VACCINE is sponsored in many
countries as the
great WONDER to combat HPV and continues
to be used by governments that maybe even knowing
the damage they cause, implement them in the
population." Here we have to think about the
motivation of them to do this? money? ignorance?
complicity? These questions are left to the
audience.
MASSIVE STERILIZATION IN KENYA WITH TETANUS VACCINE:
===================================================
On November 6, 2014, a "scandal" in
KENYA explodes in relation to the
VACCINE against the
TETANUS,
where several doctors found in several studies
of this VACCINE evidence of the existence
within them of HGC HORMONE (human
chorionic gonadotrophin), the which was developed by
WHO in the year 1.992.
This was denounced by several Doctors under
the concept that a
MASSIVE STERILIZATION was being
implemented through these
VACCINES, the
HGC in conjunction
with traces of the tetanus vaccine would
create antibodies that would prevent pregnancy in
the girls, causing a spontaneous ABORTION.
Vaccine was supplied to 2.3 million girls and women
of childbearing age.
Today, September 17, 2017, the debate on this
case was reopened where the government
is accused of having implemented the
VACCINE in the years 2.014 and 2.015,
showing that 1/3 of the vials contain HGC, a hormone
associated with the control of birth rate among
girls and women between the ages of
14 and 49 years.
It is said that between 50,000 and 500,000 women
and girls were sterilized with this
method. And I quote:
".... This is the greatest crime against humanity
ever committed against the women of
Kenya and the most devilish attempt of social
engineering .... "
THE NEW VACCINE AGAINST LYME DISEASE VL15:
==========================================
This topic and explain it to you extensively and you
can read it here:
NEW VACCINE VL15 OF VALNEVA AGAINST THE LYMD
DISEASE , ANOTHER FRAUD MORE?
I return to talk about this topic because I find the
information that the FDA is accelerating the step
for the approval of this
"NEW VACCINE" against the
LYME DISEASE or
CHRONIC ERYTHEMA MIGRANS, produced by
the SPIROCHETA BORRELIA BURGDORFERI and transmitted
by a TICK genus IXODES.
In case you did not know, 300,000 new cases
of this disease are reported annually in the
UNITED STATES, DOUBLE cases of
breast cancer and 6 times more than
cases of
AIDS.
In the year 1.998 a
VACCINE
was approved under the name of
LYMErix based on the surface antigen
OspA of BORRELIA, causal agent, which was
a tremendous disaster, said
VACCINE had to be removed from the market in
the year 2.002 by the numerous secondary
effects, diseases and
DEATH caused by it.
Subsequent studies have determined that this
OspA antigen
has the ability to "shoot" the person's
immune system. OCCASIONING
THE SAME DISEASE that "SUPPOSED" will
prevent the vaccine, and other autoimmune diseases
such as
ARTHRITIS, LUPUS and others. REALLY
the
Ospa Antigen IS AN ENDOTOXIN that causes
IMMUNOSUPPRESSION and MULTISYSTEM NEUROLOGICAL
DISEASES.
Here comes the big question? Was the
laboratory manufacturer of this event aware? or
concealed it to earn a few bucks and quiet the
NEED OF THE POPULATION of a cure against this
disease that was increasing alarming, all under the
support of the FDA.
The end result was catastrophic, you can read here
THE HISTORY OF SOME VICTIMS OF THE DISEASE OF LYME,
but not everything was there.
Today 2,017, 15 years after
LYMErix the increase to
300,000 new cases per year of
LYME DISEASE, A French pharmaceutical
called VALNEVA comes with a
"SUPPOSED" NEW VACCINE, which is
nothing more than a
"CLON" of the old LYMERIX because it is
also based on the
OspA antigen, if this is so, there is
nothing left to think but three things:
1.) THE NEW VACCINE WILL BE
ANOTHER FAILURE AND FRAUD.
2.) NEW CASES OF AFFECTED WITH
MULTISYSTEM, NEUROLOGICAL AND
AUTOIMMUNE DISEASES, AND PROBABLY
DEATHS WILL BE PRODUCED IN VACCINATED PEOPLE.
3.) IF IT IS APPROVED
IT WILL NOT LAST A LONG TIME IN THE MARKET,
MAYBE WITH THE PROFITS COVER COST EXPENSES.
HEPATITIS B VACCINE (ENGERIX-B):
=============================
The VAERS and PubMed system between the years
(1966-2003) were searched for autoimmune conditions
and the following diseases were reported
following vaccination against
HEPATITIS B:
1.) ARTHRITIS.
2.) RHEUMATOID ARTHRITIS.
3.) MYELITIS.
4.) OPTICAL NEURITIS.
5.) GUILLAIN BARRE SYNDROME (GBS).
6.) MULTIPLE SCLEROSIS (EM).
7.) VASCULITIS.
8.) SUDDEN CHILD DEATH.
9.) FATIGUE.
10.) TROMBOCYTOPENIA / PANCITOPENIA.
11.) GLOMERULONEFRITIS.
12.) ANAFILAXIS
13.) NEUROMUSCULAR DISORDERS.
14.) SYSTEMYC ERYTHEMATOUS LUPUS (LES).
15.) CHRONIC FATIGUE SYNDROME.
16.) IDIOPATHIC TROMBOCYTOPENIC PUPURA.
17.) LICHEN PLANUS: Dermatological disease, perhaps
the most reported side effect.
Not to leave the issue unfinished comes a NEW
VACCINE AGAINST HEPATITIS B in youngsters from
the age of 18 years by the pharmaceutical DYNAVAX,
is called
HELIPSAV-B (TM) which is based on the
surface antigen of the hepatitis virus B, according
to the studies this vaccine would be superior to the
already known ENGERIX-B (HEPTAVAX-B) because
"SUPPOSED" offers greater protection, with
fewer doses (2), ENGERIX-B are three doses at 0,
1, and 6 month.
The FDA in this case just delayed the approval of
said VACCINE by November of 2,017 and would be
in the market by the beginning of 2.018. THE MAIN
OBSTACLE for its approval is the side effect of
the same on the
HEART and the production of
HEART ATTACK, for that reason the FDA
again delays the approval of the same, hoping that
the company SHOW studies after its commercialization
that demonstrate security with respect to this
sensitive subject.
In case you did not know,
DYNAVAX HELIPSAV-B has been rejected for
approval by the FDA on two (2) previous occasions
in year 2,013
and year 2,016, this is its third
attempt. In one of them the same company abandoned
on its own account the attempted approval claiming
that they were not sure of their RELIABILITY.
This caused a fall in the DYNAVAX STOCK EXCHANGE
by failing in its second attempt to approve, and
I REPEAT the reason for the delay in this
approval was that in the PREVIOUS STUDIES with this
VACCINE there were
14 cases of
infarct to the myocardium.
The VACCINE has not yet been APPROVED and a
profit of
290 MILLION dollars is already projected for
the year 2.026 ... Draw your conclusions.
AH1N1 FLU VACCINE (PANDEMRIX)
=============================
In Germany, a study was conducted between
November 2.009 and December 2.010 to evaluate the
PANDEMRIX vaccine against influenza AH1N1 and its
relationship to
GUILLAIN BARRE SYNDROME and its
FISHER SYNDROME variant, which contains
ASO3 adjuvant. There were reports of
351 Hospitals with 676 GB / FS cases.
Of the 676 cases reported 30 occurred between the
first 150 days after vaccination against the AH1N1
FLU and the
GLOBAL RESULT of the investigation was that
there was a
higher incidence of
GUILLAIN BARRE SYNDROME (GBS) and its variant
FISHER SYNDROME (FS) associated with the
VACCINATION against the AH1N1 FLU with the
PANDEMRIX VACCINE.
CONJUGATED VACCINES: HAEMOPHILUS INFLUENZAE TYPE
B (Hib), ALONE OR WITH OTHERS:
=====================================================================
To end this hot topic I bring you the adverse
reactions AND DEATHS reported by the VAERS system of
vaccines against Haemophilus Influenzae type B
(hiB): Since June 2,012,
12,140 serious adverse events have been
reported to the Vaccine Adverse Event Reporting
System (VAERS) in relation to
all Hib VACCINES combined.
The majority of this number were children younger
than 3 years (11,278). Serious reactions
included
471 DEATHS and such things as
ANAPHYLACTIC REACTIONS, ASTHMA, PNEUMONIA,
CONVULSIONS, NON-INFECTIOUS ENCEPHALITIS, PERIFERIC
NEUROPATHY, ACUTE PANCREATITIS, GUILLAIN BARRE
SYNDROME, SEPSIS, AND BRAIN EDEMA.
ADVERSE EFFECTS BY VACCINE:
=============================
ACTHIB: (Haemophilus b Conjugate Vaccine (Tetanus
Toxoid Conjugate) approval 1.993.
=======
1.) Sensitivity.
2.) erythema.
3.) induration.
4.) fever.
5.) irritability.
6.) drowsiness.
7.) anorexia.
8.) diarrhea.
9.) vomiting.
when combined with the reconstituted
DTP vaccine, adverse reactions included:
1.) pain on palpation.
2.) erythema.
3.) induration.
4.) fever.
5.) irritability.
6.) drowsiness.
7.) anorexia.
8.) diarrhea.
9.) persistent crying.
10.) hypotensive / hypotensive episode (which is
consistent with the HHE rate observed with DTP
ALONE, vaccination)
HIBERIX: (Haemophilus b Conjugate Vaccine (Tetanus
Toxoid Conjugate) approval 2.009
======
When co-administered with
DTaP-HBV-IPV:
1.) REDNESS.
2.) PAIN AND SWELLING AT THE SITE OF INJECTION.
3.) FEVER.
4.) IRRITABILITY.
5.) LOSS OF APPETITE.
6.) RESTLESSNESS.
7.) DROWSINESS.
8.) DIARRHEA.
9.) VOMITING.
Postmarketing adverse events included:
1.) EXTENSIVE INFLAMMATION OF THE VACCINATED LIMB.
2.) ANAPHYLACTIC REACTIONS.
3.) ANGIOEDEMA.
4.) SEIZURES.
5.) HYPOTONIC- HYPORESPONSIVE EPISODE.
6.) SYNCOPE.
7.) APNEA.
8.) RASH.
9.) URTICARIAL.
10.) SOMNOLENCE.
PEDAVAXHIB: (Haemophilus b Conjugate Vaccine
(Tetanus Toxoid Conjugate)
============
Adverse events during clinical trials INCLUDED:
1.) irritability.
2.) drowsiness.
3.) pain / pain by injection.
4.) erythema.
5.) Swollen induration.
6.) Unusual acute crying, prolonged crying (more
than 4 hours).
7.) diarrhea.
8.) vomiting.
9.) crying.
10.) pain.
11.) otitis media.
12.) upper respiratory tract rash and infection
Potential adverse events may include the
early onset of
Hib disease and GUILLAIN-BARRE SYNDROME; in
subsequent marketing, reported adverse events
included:
1.) lymphadenopathy.
2.) angioedema.
3.) febrile convulsions.
4.) abscess at the injection site.
PENTACEL:(Diphtheria and Tetanus Toxoids and
Acellular Pertussis Adsorbed, Inactivated Poliovirus
and Haemophilus b Conjugate (Tetanus Toxoid
Conjugate) Vaccine. APPROVAL: 2.008
=========
1.) Redness.
2.) swelling.
3.) sensitivity at the injection site.
4.) increase in the circumference of the arm (dose
4).
5.) fever.
6.) lethargy.
7.) inconsolable crying.
8.) irritability.
9.) episodes of hypointensive hypotonia.
10.) seizures.
11.) febrile convulsions.
12.) bronchiolitis.
13.) dehydration.
14.) pneumonia.
15.) gastroenteritis.
16.) asthma.
17.) pneumonia.
18.) encephalopathy.
19.) four deaths attributed to asphyxia.
20.) Cranioencephalic trauma.
21.) sudden infant death syndrome (SIDS).
22.) neuroblastoma.
in postmarketing, reported adverse events included:
1.) cyanosis.
2.) vomiting.
3.) diarrhea.
4.) Extensive swelling of the injected limb,
including swelling involving Adjacent joints.
5.) Invasive Hib disease (classified as vaccine
failure).
6.) rash.
7.) urticaria.
8.) meningitis.
9.) rhinitis.
10.) viral infection.
11.) decreased appetite.
12.) drowsiness, HHE, depressed level of
consciousness.
13.) screams.
14.) apnea.
15.) cough.
16.) erythema.
17.) discoloration of the skin and pallor.
In relation to the PENTACEL vaccine, the FDA
reported:
775 adverse reactions,
177 of which were considered serious,
including
26 DEATHS, between June 20, 2008 (when
Pentacel was licensed) and October 31, 2009.
Deaths were attributed to SIDS (12 cases),
congenital / genetic conditions, respiratory
infections, positional asphyxia, anoxic
encephalopathy, cardiac arrest of undetermined
etiology, dilated and hypertrophic cardiomyopathy,
two deaths of indeterminate cause, one death without
available records and two deaths with pending
information .
MENHIBRIX: X (Meningococcal Groups C and Y and
Haemophilus b Tetanus Toxoid Conjugate Vaccine)
APPROVAL: 2.012
==========
1.) Redness.
2.) swelling and pain at the injection site.
3.) irritability.
4. drowsiness
5.) loss of appetite.
6.) fever.
7.) syncope.
For more serious reactions, such as
NERVOUS SYSTEM DISORDERS and other
serious events, the manufacturer referred to
reactions reported by the use of Hiberix instead of
his own vaccine. It is not known whether
MenHibrix can cause
FETAL HARM
when given to a pregnant woman or if it may
affect reproduction capacity.
CONCLUSIONS:
=============
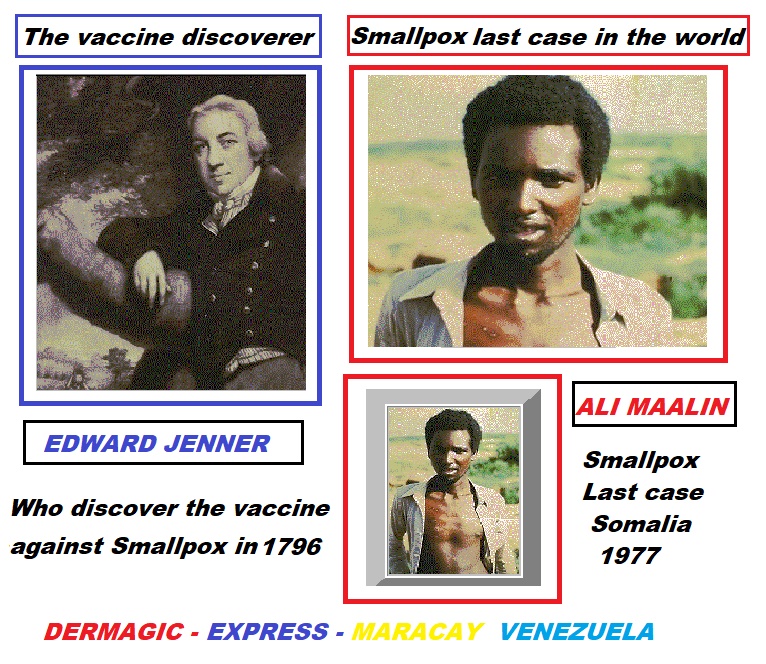
1.) The man has made great discoveries and
through some of his
VACCINES
such as the case of
smallpox
EDWARD JENNER who in 1.796 passed to
History when discovering the vaccine that put an end
to that disease in ALL THE WORLD, and whose last
case was registered in SOMALIA, 1,977 in the man
ALI MAALIN who also WAS IMMORTALIZED as
the last case.
2.) Great discoveries have been made regarding
VACCINES many of which are necessary to
avoid EPIDEMIC, it is true, such is the case of
HEPATITIS A and B.
3.) But also some vaccines have been great
failures, such as the vaccine against LYME
DISEASE
(LYMErix) and HPV
(GARDASIL AND CERVARIX) that far from
preventing have caused more disease and deaths.
4.) Many of the VACCINES contain adjuvants
such as
ALUMINUM AND
THIMEROSAL (MERCURY) which are
triggers of immunological reactions, and instead of
preventing, it triggers disease.
5.) Other vaccines for their
BAD DESIGN,
produce severe side effects and some of them are
linked to proven cases of
AUTISM.
6.) The administration of the
VACCINES SHOULD be in well-studied,
longer periods of time, not take a child and put
5 or 6 vaccines in a single session. In
my opinion that is ONE OF THE GREAT FAULTS.
7.) The United States court has called the
so-called resolution 327 where it orders the
mandatory vaccines, discussed in Congress on
May 16, 2017, alleging the safety of vaccines
and that these do not cause side effects.
They are wrong. The vaccines are
necessary, it is true, but many of them
can KILL you or SICK you or your childs for ever and
ever.
8.) It is your decision to TAKE THE
VACCINE,
but in the United States, the government has just
put in jail for 7 days a mother (Rebbeca
Bredow) who refused to put several shots to her son
thinking about his health.
9.) In JAPAN, the government eliminated the
mandatory of some
VACCINES, and banned others, and is one
of the countries with the highest health index in
children.
10.) The Social Networks have succeeded in
expanding in the world community the current
problem of
VACCINES, and the rejection of the
population towards them.
11.) The most questioned
VACCINE of all is the MMR against
Rubella measles, and mump, and to which
it has been associated cases of
AUTISM, death and other diseases, in fact
it is the most rejected today.
All this I wrote is the
CRYSTAL reality, and is well supported,
as always DERMAGIC EXPRESS tells you the truth,
there is no invention, read the BIBLIOGRAPHIC
references.
Greetings to all
Dr. José Lapenta
EDITORIAL ESPAÑOL
=================
Bienvenidos una vez mas todos los amigos, médicos,
dermatólogos, gente alrededor del mundo, lectores
del DERMAGIC EXPRESS, hoy una vez más hablando del
controversial tema de las VACUNAS, bajo el
nombre NO-VAXX, SIEMPRE DI: NUNCA DE NUEVO.
Para comenzar les diré una vez más, que esta no es
la primera vez y quizá no será la ultima, que hablo
sobre este tema que hoy día se ha convertido en uno
de los más discutidos en la comunidad mundial, tema
QUE HA REVIVIDO por los
efectos ADVERSOS y
MUERTES ocasionadas por las VACUNAS CONTRA EL
VPH, GARDASIL y CERVARIX, los casos de AUTISMO Y
muerte provocados por algunas VACUNAS, y para
derramar la gota del vaso, EL próximo
NACIMIENTO DE LA "NUEVA" VACUNA VL15 de
la farmacéutica Valneva CONTRA la
ENFERMEDAD DE LYME.
Primeramente hay que dividir a la población en
tres grupos: 1.) Aquellos
que no saben o desconocen el problema de las
VACUNAS o no les interesa. 2.)
Los "VAXERS" aquellas personas que
están de acuerdo con la VACUNAS. Y 3.)
Los llamados
ANTI-VAXERS, aquellas personas que por
una u otra razón
se oponen a las VACUNACIONES,
individuales y masivas, la mayoría de ellas
porque algún familiar o hijo sufrió EFECTOS
ADVERSOS o MUERTE por alguna VACUNA, o
personalmente quedo INCAPACITADO luego de
alguna vacunación.
Para darle TOTAL credibilidad a este post voy a
comenzar colocándote los
EFECTOS ADVERSOS Y
MUERTES reportados por el SISTEMA DE
NOTIFICACION DE EVENTOS ADVERSOS (VAERS) En los
estados Unidos sobre la
VACUNA CONTRA EL SARMPION, RUBEOLA Y
PAROTIDITIS, la muy conocida MMR.
VACUNA MMR (SARAMPION PAROTIDIS Y RUBEOLA):
============================================
Se reportaron
75.000 mil efectos adversos que
incluyen:
EN GENERAL: vómitos, diarrea, anafilaxia, dolor
de oído, sordera nerviosa, diabetes, artritis,
mialgia, encefalitis, convulsiones febriles,
neumonía y
muerte; EN DETALLE:
1.)
78 muertes
2.)
85 casos de sordera
3.)
48 casos de disminución del contacto visual
4.)
92 casos de retraso en el desarrollo
5.)
855 casos reportados de autismo
6.)
116 casos de discapacidad intelectual
7.)
401 informes de trastornos del habla
8.)
276 informes de pérdida de conciencia
9.)
143 casos de encefalitis
10.)
74 casos de meningitis
11.)
111 casos de síndrome de Guillain-Barré.
12.)
692 casos de alteración de la marcha (no
poder caminar normalmente)
13.)
748 casos de hipoquinesia (pérdida parcial o
total del movimiento muscular)
14.)
653 informes de hipotonía (pobre tono
muscular)
15.)
4874 informes de convulsiones, incluidas las
convulsiones febriles y las convulsiones tónicas
clónicas
16.)
1576 CASOS DE CELULITIS (INFECCION DE PIEL
POTANCIALMENTE GRAVE).
En algunos casos, la VACUNA ha causado las mismas
enfermedades que se supone que previene, con los
siguientes datos informados al VAERS:
17.)
147 CASOS DE SARAMPION.
18.)
384 CASOS DE PAROTIDITIS.
19.)
29 CASOS DE RUBEOLA.
Estos números no son EXACTOS pues el reporte es del
1 al 10% de las reacciones, por lo tanto pueden ser
mayores.
JAPON PROHIBE VACUNA MMR (RUBEOLA, PAROTIDITIS Y
SARAMPION):
=============================================================
Sobre esta VACUNA MMR quizá no sabías que EL
GOBIERNO JAPONES, la prohibió en el año 1.993
(hace 24 años) después que 1.800.000
niños fueron vacunados con 2 tipos MMR y un alto
porcentaje presento signos de
MENINGITIS NO VIRAL y OTRAS RECACCIONES
ADVERSAS, entre las que destacan
3 NIÑOS MUERTOS, y
8 CON DISCAPACIDADES PERMANENTES: SORDERA,
CEGUERA y PERDIDA DEL CONTROL DE LAS EXTREMIDADES.
Además cito textualmente:
"....De las 3,969 reclamaciones de INDEMNIZACION
MEDICA relacionadas con las vacunas en los últimos
30 años, una cuarta parte lo hicieron los afectados
por la vacuna combinada contra el sarampión, las
paperas y la rubéola..."
En 1.999 JAPON considero volver a utilizar la VACUNA
pero prefirió mantener LA PROHIBICION y
utilizar
individualmente las VACUNAS contra SARAMPION,
PAROTIDITIS Y RUBEOLA. El costo es más elevado,
pero vale la pena. Los efectos SECUNDARIOS Y MUERTES
son menores.
GARDASIL Y CERVARIX (VACUNAS CONTRA EL VPH):
============================================
De la vacuna GARSASIL y CERVARIX y sus efectos
adversos ya te he hablado bastante y los links los
tienes acá:
1.)
PROHIBAN LA VACUNA GARDASIL Y CERVARIX
CONTRAL EL VPH, VIRUS DEL PAILOMA HUMANO.
2.)
VACUNA GARDASIL CONTRA EL VPH & MERCK DEMANDADOS
EN COLOMBIA, SINDROME POST GARDASIL Y CERVARIX POST
VACUNACION.
JAPON PROHIBE VACUNAS GARDASIL Y CERVARIX (VPH)
EN 2.013
========================================================
Pero lo que quizá no sabias tu que te colocas
del lado de los "VAXERS" es que JAPON una
potencia mundial
PROHIBIO, "BANEO" la
VACUNA GARDASIL en el año 2.013 debido al
reporte de aproximadamente
2.000 EFECTOS SECUNDARIOS, cientos de los
cuales fueron GRAVES.
Reportaron los Japoneses en aquella epoca:
CONVULSIONES, DAÑO CEREBRAL, CEGUERA, PARALISIS,
PROBLEMAS DEL HABLA, PANCREATITIS, PERDIDA DEL HABLA
A CORTO PLAZO, MUERTE y OTROS. Y vuelvo
a citar textualmente
"...Desde que el gobierno comenzó a ofrecer
inyecciones de VPH para niñas, se notificaron
1.968 eventos adversos, de los cuales
358 fueron evaluados como serios por un
comité JMLHW. Los padres comenzaron a llamar al
ministro de salud del país y le proporcionaron
videos en los que las niñas que habían recibido
la vacuna contra el VPH sufrían
alteraciones de la marcha, tics corporales y
convulsiones. En otros casos,
muchas niñas a las que se les inyectó la vacuna
cayeron al suelo, lesionándose la cabeza o la
cara y algunas fracturándose la mandíbula o los
dientes..."
Y aun asi esta "VACUNA sigue siendo
patrocinada en muchos paises como
la gran MARAVILLA para combatir el VPH.
y sigue utilizandose por gobiernos que quiza
aun sabiendo el daño que causan, las implementan en
la poclacion. Aqui habria que pensar cual es la
motivacion de ellos para hacer esto ? dinero ?
desconocimiento? complicidad ? Estas preguntas
se las dejo a la audiencia.
ESTERILIZACION MASIVA EN KENYA CON LA VACUNA CONTRA
EL TETANO:
===============================================================
El 6 de noviembre de 2014 "explota" en
KENYA un escándalo en relación a la
VACUNA contra el
TETANOS,
donde varios doctores encontraron en
varios estudios de esta VACUNA evidencia de la
existencia
dentro de ellas de la HORMONA HGC
(gonadotrofina corionica humana), la cual fue
desarrollada por la OMS en él año 1.992.
Esto fue denunciado por varios Doctores bajo
el concepto que se estaba implementando una
ESTERILIZACION MASIVA a través de estas
VACUNAS,
la
HGC en conjunción con los
rastros de la vacuna contra el tétano
crearía anticuerpos que evitarían el embarazo en
las niñas, provocando UN ABORTO espontaneo. La
VACUNA fue suministrada a 2.3 millones de niñas y
mujeres en edad fértil.
Hoy, 17 de Septiembre de 2.017 se volvió a
abrir el debate sobre este caso donde se acusa al
gobierno de haber implementado la
VACUNA en los
años 2.014 Y
2.015 exponiéndose que 1/3 de los viales
contienen HGC, una hormona asociada al control de la
natalidad en niñas y mujeres en edad comprendida
entre 14 y 49 años de edad.
Se dice que entre 50.000 y 500.000 mil mujeres y
niñas quedaron esterilizadas con este
método. Y cito textualmente:
"...."Este es el mayor crimen contra la humanidad
jamás cometido contra las mujeres de
Kenia y el intento más diabólico de
ingeniería social...."
LA NUEVA VACUNA CONTRA LA ENFERMEDAD DE LYME
VL15:
====================================================
Este tema ya te lo explique ampliamente y puedes
leerlo aquí:
NUEVA VACUNA VL15 DE VALNEVA CONTRA LA ENFERMEDAD
DE LYME, OTRO FRAUDE MÁS?
Vuelvo a hablar sobre este tema porque me encuentro
con la información que LA FDA está acelerando el
paso para la aprobación de esta
"NUEVA VACUNA"
contra la
ENFERMEDAD DE LYME
o ERITEMA CRONICO MIGRANS,
producida por la ESPIROQUETA BORRELIA BURGDORFERI y
transmitida por una GARRAPATA del género IXODES.
Por si no lo sabías se reportan anualmente en los
ESTADOS UNIDOS 300.000 mil casos nuevos
de esta enfermedad, EL DOBLE de los
casos de cáncer de seno y 6 veces más que los
casos de SIDA.
En el año 1.998 se aprobó una
VACUNA bajo el nombre de
LYMErix basada en el
antígeno de superficie OspA de la
BORRELIA, agente causal, la cual fue un tremendo
desastre, dicha VACUNA tuvo que ser sacada
del mercado en el año 2.002 por los innumerables
efectos secundarios, enfermedades y MUERTE
ocasionados por la misma.
Estudios posteriores determinaron que este
antígeno OspA tiene la capacidad de
"disparar" el sistema inmune de la persona
OCASIONANDO
LA MISMA ENFERMEDAD que
"SUPUESTAMENTE" te va a evitar la
vacuna, y además otras ENFERMEDADES AUTOINMUNES
como
ARTRITIS, LUPUS y otras, y esto se
debe a que REALMENTE el
Antígeno Ospa ES UNA ENDOTOXINA que
ocasiona INMUNOSUPRESION y ENFERMEDADES NEUROLOGICAS
MULTISISTEMICAS.
Aquí viene la gran pregunta ? Conocía el
laboratorio fabricante de este evento ? o lo
encubrió para ganar unos cuantos dólares y acallar
la NECESIDAD DE LA POBLACION de una cura
contra esta enfermedad QUE estaba aumentando
alarmante, todo bajo el apoyo de la FDA.
El resultado final fue catastrófico, tú puedes leer
acá
LA HISTORIA DE ALGUNAS VICTIMAS DE LA ENFERMEDAD
DE LYME, pero no todo quedo allí.
Hoy 2.017, 15 años después de
LYMErix el aumento a
300.000 mil casos nuevos por año de la
ENFERMEDAD DE LYME, Una farmacéutica
Francesas llamada VALNEVA se viene con una
SUPUESTA NUEVA VACUNA, la cual no es más
que un
"CLON" de la vieja LYMERIX pues
también está basada en el
antígeno OspA, si esto es así, aquí no
queda otra que pensar 3 cosas:
1.) LA NUEVA VACUNA SERA
OTRO FRACASO MAS.
2.) SE PRODUCIRAN
NUEVOS CASOS DE AFECTADOS CON ENFERMEDADES
MULTISISTEMICAS, NEUROLOGICAS Y AUTOINMUNES
EN LOS VACUNADOS, Y PROBABLEMENTE
MUERTES.
3.) SI ES APROBADA
NO DURARA MUCHO TIEMPO EN EL MERCADO, QUIZA
CON LAS GANANCIAS CUBRAN LOS GASTOS DEL COSTO.
VACUNA CONTRA LA HEPATITIS B (ENGERIX-B):
========================================
El sistema VAERS y PubMed entre los años
(1966-2003) fueron buscados por condiciones
autoinmunes y se reportaron las siguientes
enfermedades, luego de la vacunación contra
HEPATITIS B:
1.) ARTRITIS.
2.) ARTRITIS REUMATOIDE.
3.) MIELITIS.
4.) NEURITIS OPTICA.
5.) SINDROME DE GUILLAIN BARRE (SGB).
6.) ESCLEROSIS MULTIPLE (EM).
7.) VASCULITIS.
8.) MUERTE SUBITA INFANTIL.
9.) FATIGA.
10.) TROMBOCITOPENIA / PANCITOPENIA.
11.) GLOMERULONEFRITIS.
12.) ANAFILAXIS
13.) DESORDENES NEUROMUSCULARES.
14.) LUPUS ERITEMATOSO SISTEMICOL(LES).
15.) SINDROME DE FATIGA CRONICA.
16.) PUPURA TROMBOCITOPENICA IDIOPATICA.
17.) LIQUEN PLANO: Enfermedad dermatológica, quizá
el efecto secundario mas reportado.Para no dejar
el tema inconcluso se viene UNA NUEVA VACUNA
CONTRA LA HEPATITIS B en jóvenes a partir de
los 18 años por la farmacéutica
DYNAVAX, se denomina
HELIPSAV-B (TM) la cual ESTA BASADA
EN EL ANTIGENO DE SUPERFICIE DEL VIRUS DE la
hepatitis B, según los estudios esta vacuna
sería superior a la ya conocida
ENGERIX-B (HEPTAVAX-B) porque
"SUPUESTAMENTE" ofrece una mayor protección, con
menos dosis (2), ENGERIX-B son tres dosis al 0, 1, Y
6to mes.
La FDA en este caso acaba de retrasar la
aprobación de dicha
VACUNA para noviembre de 2.017 y estaría
en el mercado para comienzos del 2.018. EL
PRINCIPAL OBSTACULO para su aprobación es el
efecto secundario de la misma sobre el
CORAZON
y la producción de
INFARTO, por ello la FDA vuelve a
retrasar la aprobación de la misma, esperando que la
empresa MUESTRE estudios posteriores a su
comercialización que demuestren seguridad con
respecto a este tema delicado.
Por si no lo sabías DYNAVAX HELIPSAV-B ha sido
rechazada para su aprobación por la FDA en dos (2)
ocasiones previas en él
año 2.013 y el
año 2.016, este es su tercer intento. En
uno de ellos la misma compañía abandono por su
propia cuenta el intento de aprobación alegando que
no estaban seguros de su CONFIABILIDAD.
Esto provoco una caída en la BOLSA DE VALORES DE
DYNAVAX al fallar en su segundo intento de
aprobación,
Y REPITO la razón del retraso en esta
aprobación fue que en que los ESTUDIOS
PREVIOS con esta
VACUNA HUBO
14 CASOS DE INFARTO AL MIOCARDIO.
La VACUNA todavía NO ha sido APROBADA y ya se
proyecta
una ganancia de 290 MILLONES DE dólares
para él año 2.026... Saque usted sus conclusiones.
VACUNA CONTRA LA GRIPE AH1N1 (PANDEMRIX):
=========================================
En Alemania se hizo un estudio entre noviembre
2.009 y diciembre del 2.010 para evaluar la
vacuna
PANDEMRIX contra la influenza AH1N1 y su
relación con el
SINDROME DE GUILLAIN BARRE (GBS) Y su
variante EL
SINDROME DE FISHER (FS), dicha vacuna
contiene el
adyuvante ASO3. Hubo reportes de 351
Hospitales con 676 casos de GB / FS.
De los 676 casos reportados 30 ocurrieron entre
los primero 150 días posterior a la vacunación
contra la GRIPE AH1N1 Y
EL RESULTADO GLOBAL de la investigación
fue que hubo
una mayor incidencia DE
SINDROME DE GUILLAIN BARRE (GBS) y su
variante
SINDROME DE FISHER (FS) asociados a la
VACUNACION contra la GRIPE AH1N1 con la VACUNA
PANDEMRIX.
VACUNAS CONJUGADAS CONTRA HAEMOPHILUS INFLUENZAE
TIPO B (Hib), SOLA O CON OTRAS:
===================================================================
Para finalizar este caliente tema te traigo las
reacciones adversas Y MUERTES reportadas por el
sistema VAERS de las
VACUNAS contra
Haemophilus Influenzae tipo B (hiB): Desde
junio de 2,012, se han notificado
12.140 eventos adversos graves al Sistema
de Informe de Eventos Adversos a Vacunas (VAERS) en
relación con todas las
VACUNAS Hib combinadas.
La mayoría de este número eran niños menores de
3 años (11,278). Las reacciones graves
incluyeron
471 MUERTES y cosas tales como
REACCION ANAFILACTICA, ASMA, NEUMONIA, CONVULSIONES,
ENCEFALITIS NO INFECCIOSA, NEUROPATIA PERIFERICA,
PANCREATITIS AGUDA, SINDROME DE GUILLAIN BARRE,
SEPSIS Y EDEMA CEREBRAL.
EFECTOS ADVERSOS POR VACUNA:
==============================
ACTHIB: (HAEMOPHILUS B Y TOXOIDE TATANICO
CONJUGADOS) APROBADA 1.993
=======
1.) Sensibilidad.
2.) eritema.
3.) induración.
4.) fiebre.
5.) irritabilidad.
6.) somnolencia.
7.) anorexia.
8.) diarrea.
9.) vómitos.
cuando se combinó con la vacuna DTP por
reconstitución, las reacciones adversas incluyeron:
1.) dolor a la palpación.
2.) eritema.
3.) induración.
4.) fiebre.
5.) irritabilidad.
6.) somnolencia.
7.) anorexia.
8.) diarrea.
9.) llanto persistente.
10.) episodio hipotónico / hipotensor (que es
consistente con la tasa de HHE observada con la
vacunación con DTP SOLA)
HIBERIX: (Haemophilus b VACUNA CONJUGADA (ToxoidE
TETANICO ConjugaDO) apprOBADA 2.009
======
CUANDO SE CO-ADMINISTRA
CON DTaP-HBV-IPV:
1.) ENROJECIMIENTO.
2.) DOLOR E HINCHAZON EN EL SITIO DE LA INYECCION.
3.) FIEBRE.
4.) IRRITABILDAD.
5.) PERDIDA DEL APETITO.
6.) INQUIETUD
7.) DESMAYO
8.) DIARREA.
9.) VOMITOS.
EVENTOS ADVERSOS POSTMERCADEO INCLUYEN:
1.) INFLAMACION EXTEBSA EN EL MIEMBRO VACUNADO.
2.) REACCIONES ANAFILACTICAS.
3.) ANGIOEDEMA.
4.) CONVULSIONES
5.) EPISODIOS HIPOTONICOS- Y DISMINUCION DE
RESPUESTAS.
6.) SINCOPE.
7.) APNEA.
8.) RASH.
9.) URTICARIA.
10.) SOMNOLENCIA
PEDAVAXHIB: (HAEMOPHILUS B Y MENINGOCOCO VACUNA
CONJUGADA)
============
Los eventos adversos durante los ensayos clínicos
INCLUYERON:
1.) irritabilidad.
2.) somnolencia.
3.) dolor / dolor por inyección.
4.) eritema.
5.) induración hinchada.
6.) llanto inusual agudo, llanto prolongado (más de
4 horas).
7.) diarrea.
8.) vómitos.
9.) llanto.
10.) dolor.
11.) otitis media.
12.) erupción e infección de las vías respiratorias
superiores
los eventos
adversos potenciales pueden incluir la aparición
temprana de la enfermedad Hib y
el síndrome de Guillain-Barre; en la
comercialización posterior, los eventos adversos
informados incluyeron:
1.) linfadenopatía.
2.) angioedema.
3.) convulsiones febriles.
4.) absceso en el lugar de la inyección.
PENTACEL: ( VACUNA CONTRA: DIFTERIA, TETANO,
PERTUSIS, POLIO Y HEAMOPHILUS (DTaP-IPV/Hib)
APROBADA 2.008
==============
1.) Enrojecimiento.
2.) hinchazón.
3.) sensibilidad en el lugar de la inyección.
4.) aumento de la circunferencia del brazo (dosis
4).
5.) fiebre.
6.) letargo.
7.) llanto inconsolable.
8.) irritabilidad.
9.) episodios de hipotonía hipointensiva.
10.) convulsiones.
11.) convulsiones febriles.
12.) bronquiolitis.
13.) deshidratación.
14.) neumonía.
15.) gastroenteritis.
16.) asma.
17.) neumonía.
18.) encefalopatía.
19.) cuatro muertes atribuidas por asfixia.
20.) traumatismo craneoencefálico.
21.) síndrome de muerte súbita del lactante (SMSL).
22.) neuroblastoma.
en la postcomercialización, los eventos adversos
informados incluyeron:
1.) cianosis.
2.) vómitos.
3.) diarrea.
4.) hinchazón extensa de la extremidad inyectada,
incluyendo Hinchazón que involucraba articulaciones
adyacentes.
5.) enfermedad Hib invasiva (clasificada como falla
de la vacuna).
6.) erupción cutánea.
7.) urticaria.
8.) meningitis.
9.) rinitis.
10.) infección viral.
11.) disminución del apetito.
12.) somnolencia, HHE, nivel de conciencia
deprimido.
13.) gritos.
14.) apnea.
15.) tos.
16.) eritema.
17.) decoloración de la piel y palidez.
En relación a la vacuna
PENTACEL la FDA informo:
775 reacciones adversas,
177 de las cuales se consideraron graves,
incluidas
26 MUERTES, entre el 20 de junio de 2008
(cuando Pintase tenía licencia) y el 31 de octubre
de 2009.
Las muertes se atribuyeron a SIDS (12 casos),
condiciones congénitas / genéticas, infecciones
respiratorias, asfixia posicional, encefalopatía
anóxica, paro cardíaco de etiología indeterminada,
miocardiopatía dilatada e hipertrófica, dos muertes
de causa indeterminada, una muerte sin registros
obtenibles y dos muertes con información pendiente.
MENHIBRIX: (VACUNA CONTRA HAEMOPHILUS B,
MENINGOCOCO C y Y,Y TETANOS CONUJUGADA) APROBADA
2.012
==========
1.) Enrojecimiento.
2.) hinchazón y dolor en el lugar de la inyección.
3.) irritabilidad.
4.) somnolencia
5.) pérdida del apetito.
6.) fiebre.
7.) síncope.
Para reacciones más graves, como
TRASTORNOS DEL SISTEMA NERVIOSO y otros
eventos graves, el fabricante se refirió a las
reacciones informadas por el uso de Hiberix en lugar
de a su propia vacuna. No se sabe si
MenHibrix puede causar
DAÑO FETAL cuando se lo administra a una
mujer embarazada o si puede afectar la capacidad de
reproducción.
CONCLUSIONES:
==============
1.) El hombre ha hecho grandes descubrimientos y
a través de algunas de sus
VACUNAS
como el caso de la
Viruela
EDWARD JENNER quien en 1.796 paso a la
Historia al descubrir la vacuna que puso fin a esa
enfermedad en TODO EL MUNDO, y cuyo último
caso fue registrado en SOMALIA, 1.977 en el
hombre
ALI MAALIN quien también QUEDO
INMORTALIZADO como el último caso.
2.) Se han hecho grandes descubrimientos en
cuanto a
VACUNAS muchas de las cuales son
necesarias para evitar EPIDEMIAS, es cierto, tal es
el caso de la HEPATITIS A Y B.
3.) Pero también algunas VACUNAS HAN SIDO GRANDES
FRACASOS, como la vacuna contra la
ENFERMEDAD DE LYME
(LYMErix) y contra el
VPH
(GARDASIL Y CERVARIX)
que lejos de prevenir han ocasionado mas
enfermedad y muertes.
4.) Muchas de las
VACUNAS
contienen adyuvantes como
ALUMINIO Y
TIMEROSAL (MERCURIO) los cuales
son desencadenantes de reacciones inmunológicas, y
en lugar de prevenir, desencadena enfermedades.
5.) Otras vacunas por su
MAL DISEÑO, producen efectos secundarios
severos y algunas de ellas están vinculadas a
probados casos de
AUTISMO.
6.) La administración de las
VACUNAS DEBERIA ser en espacios de tiempo
bien estudiados, más prolongados, no llevar a
un nino y colocarle
5 o 6 vacunas en una sola sesión. En mi
opinión ese es UNO DE LOS GRANDES FALLOS.
7.) La corte de estados Unidos tiene lista la
llamada resolución 327 donde ordena la
obligatoriedad de las vacunas, discutida en
el Congreso el 16 de Mayo 2.017 alegando la
seguridad de las vacunas y que estas no ocasionan
efectos secundarios.
Están equivocados. SON necesarias, es
cierto, pero muchas de ellas
pueden MATARTE o ENFERMAN a ti o a tus hijos por
siempre.
8.) Es tu decisión
VACUNARTE, pero en estados unidos el
gobierno
acaba de poner presa por 7 días a una madre
(Rebbeca Bredow) que se negó a colocarle varias
vacunas a su hijo pensando en su salud.
9.) En JAPON el gobierno elimino la
obligatoriedad de algunas
VACUNAS,
y PROHIBIO otras, y es uno de los paises con mayor
indice de salud en los niños.
10.) Las Redes Sociales han logrado expandir en
la comunidad mundial el problema actual de las
VACUNAS, y el rechazo de la poblacion
hacia ellas.
Todo esto que escribí es la
CRISTALINA realidad, y está bien
soportado, como siempre DERMAGIC EXPRESS te dice la
verdad, no hay invento, lee las referencias
BIBLIOGRAFICAS.
Saludos a Todos
Dr. José Lapenta
=======================================================================
REFERENCIAS BIBLIOGRAFICAS/
BIBLIOGRAPHICAL REFERENCES
=======================================================================
1.) Risk of Guillain-Barré syndrome following pandemic influenza
A(H1N1) 2009 vaccination in Germany.
2.) Clinical Features of Post-Vaccination Guillain-Barré
Syndrome (GBS) in Korea.
3.) Lichen planus secondary to rabies vaccination.
4.) Lichen planus occurring after influenza vaccination: report
of three cases and review of the literature.
5.) Lichen planus after HBV vaccination in a child: a case
report from Nepal.
6.) Lichen planus induced by hepatitis B vaccination: a new case
and review of the literature.
7.) Hepatitis B vaccination and associated oral manifestations:
a non-systematic review of literature and case reports.
8.) A case-series of adverse events, positive re-challenge of
symptoms, and events in identical twins following hepatitis B
vaccination: analysis of the Vaccine Adverse Event Reporting
System (VAERS) database and literature review.
9.) A one year followup of chronic arthritis following rubella
and hepatitis B vaccination based upon analysis of the Vaccine
Adverse Events Reporting System (VAERS) database.
10.) Hepatitis B vaccination and adult associated
gastrointestinal reactions: a follow-up analysis.
11.) Allergic reactions to Japanese encephalitis vaccine.
12.) The web and public confidence in MMR vaccination in Italy.
13.) Media coverage of the measles-mumps-rubella vaccine and
autism controversy and its relationship to MMR immunization
rates in the United States.
14.) Vaccine Adverse Events: Separating Myth from Reality.
15.) Refusal to Vaccinate Child Gets Mom Jail Time: A Deeper
Analysis
16.) Why Japan banned MMR vaccine
17.) Japanese Government Continues to Ban the MMR Vaccine
18.) A mass sterilization exercise’: Kenyan doctors find anti-fertility
agent in UN tetanus vaccine
19.) Raila Odinga: Tetanus vaccination is a mass sterilization
on women
20.) Infant Dies Following 5 Vaccine Doses
21.) NEW LYME VACCINE COMING SOON. CAVEAT EMPTOR––BUYER BEWARE!
22.) SIDE EFFECTS IN YOUNG GIRLS TAKE GARDASIL OUT FROM JAPANESE
MARKET
23.) Vaccines do NOT cause injuries, according to House
Resolution 327
24.) H. RES. 327
25.) Neurological complications of vaccination with outer
surface protein A (OspA).
26.) Spirochetal Lipoproteins and Immune Evasion
27.) Does a New Hepatitis Vaccine Cause Heart Attacks?
28.) FDA extends review on Dynavax's Heplisav-B, but management
remains confident
29.) Can Hib Vaccine Cause Injury?
==================================================================
===================================================================
1.) Risk of Guillain-Barré syndrome following pandemic
influenza A(H1N1) 2009 vaccination in Germany.
===================================================================
Pharmacoepidemiol Drug Saf. 2014 Nov;23(11):1192-204. doi:
10.1002/pds.3638. Epub 2014 May 10.
Prestel J1, Volkers P, Mentzer D, Lehmann HC, Hartung HP,
Keller-Stanislawski B; GBS Study Group.
Collaborators (370)
Gerber J, Heyden M, Klötzsch C, Linster E, Langel S, Bößenecker
W, Bamberg C, Gerlach R, Reckhardt M, Borak P, Braun H, Harzheim
M, Isenhardt K, Bader P, Glocker F, Reimers CD, Riesterer-Hemm
G, Trabold R, Homberg V, Klee R, von Rosen F, Daffertshofer M,
Hofler D, Hofstadt U, Baumsteiger C, Dreger J, Stachulski F,
Harms H, Kauert A, Haas J, von Brevern M, Völzke E, Ducke F,
Böhme H, Koennecke HC, Ecke A, Koennecke HC, Bähr D, Nabavi DG,
Hopmann D, Porz D, Neumayer B, Müller T, Kitzrow M, Börnke C,
Schröder A, Kowalski T, Günther J, Boeckler D, Reinshagen A,
Sarholz M, Böhm KD, Weiland T, Kuhlmann RJ, Heide W, Heider S,
Schüler O, Schepelmann K, Claus D, Kunesch E, Yaretskyy G,
Spieker S, Jung S, Spitzer C, Busch A, Schneider H, Machetanz J,
Grehl H, Nolden-Koch M, Wilmsen H, Lehmann H, Seitz R, Griese M,
Schmitt HM, Dietze C, Holz J, Leinisch E, Derfuß T, Linker R,
Reinhardt FM, Krämer M, Koeppen S, Gerhard H, Bauer H, Stolze H,
Jost V, Steinmetz H, Schütz H, Böhm J, Rauer S, Klotz JM,
Schneider A, Biemann M, Blaes F, Krämer H, Guthke K, Schmidt H,
Krumpolt H, Schiess D, Roth M, Fuchs HH, Schneider E, Wohlfarth
K, Müller T, Sick W, Schwandt D, Wellach I, Töpper RF, Koehler
J, Rosenkranz T, Knepel S, Duwe T, Stiller M, Rieke K, Baas H,
Brunotte P, Tümmler J, Heidenreich F, Stangel M, Stift T,
Ringleb P, Kaendler S, Humbroich K, Plöger H, Sitzer M, Dieter
J, Dietz M, Eicke M, Ochs G, Güldenring A, Hagemann G, Zinke J,
Fetter M, Herath H, Escheu G, Stingele R, Berthold A, Schmitt E,
Haupt WF, Petereit HF, Limmroth V, von Giesen HJ, Kirsch B,
Heckmann JG, Beuche W, Schattenfroh C, Tampier-Pohl C, Friedl R,
Oberwittler C, Schlenker M, Trillenberg P, Abushammala A,
Schabet M, Eßer M, Thümen A, Lins H, Vielhaber S, Bayerl J,
Tackenberg B, Hachgenei A, Meisenheim GK, Philipps J, Tings T,
Franz O, Jauß M, Berthele A, München TU, Hupfer W, Nagi M,
Dziewas R, Kusch W, Lobenstein S, Fischer V, Syed N, Bitsch A,
Jahnke U, Allendörfer J, Dietrich W, Görtzen A, Stark E, Wenning
W, Neumann F, Petrick M, Kaiser R, Niehoff T, Deymann R,
Hartmann R, Christe W, Görlitz C, Hotz M, Buchner H, Balzer K,
Braune HJ, Kindl HJ, Leschnik O, Lohner H, Zettl U, Kiefer R,
Krauth S, Hansberg T, Matrisch H, Vetter T, Schepelmann K,
Schade B, Nguento A, Fortwängler T, Hartnack F, Neuhaus O,
Wennrich M, Leopold HC, Tebben J, Polzer U, Sieb P, Huss GP,
Kuhl V, Gawlitza M, Freudenberger T, Rieder G, Schröder K, Bös
M, Krüger T, Rechlin M, Bufler J, Angerer M, Möller P, Eppinger
B, Dömges F, Albrecht P, Kotterba S, Stolz E, Schmidt N,
Trottenberg T, Feige P, Hufschmidt A, Svrakova L, Haensch CA,
Kastrau F, Schmidt P, Reiners K, Weinmann E, Merkelbach S,
Bachhuber A, Hermann W, Häusler M, Toth M, Weiß BM, Stein D,
Klepper J, Hirsch T, von Moers A, Brandes H, Botsch M, Köhler C,
Franke I, Kössel H, Kauffmann B, Schwalm H, Kirschstein M,
Tribukait U, Mandl M, Böhmann H, Schaetz K, Hebing B, Steinert
M, Eichholz S, von Lilien-Waldau T, Karenfort M, Kretzschmar B,
Trollmann R, Schmiedel G, Dördelmann M, Kieslich M, Mause U,
Grüber C, Korinthenberg R, Heubner G, Mattes J, Radlow U, Repp
R, Klinge J, Genseke R, Gsinn S, Gebhardt B, Papsch M, Mutlak S,
Brockmann K, Koch G, Mandelkow F, v Blanckenburg P, Bertram U,
Vieker S, Peltner HU, Sander M, Shamdeen MG, Nowka S, Koch W,
Walkenhorst H, Rubens T, Westerbeck K, Rübo J, Wiater A, Pflumm
K, Bensch J, Engelhardt H, Deja M, Borte M, Tibussek D, Bosse H,
Rinschen K, Härtel C, Gaude-Wagener M, Beyer U, Pädiatrie A,
Peters H, Kowalzik F, Seipelt P, Zippel S, Pargac KN, Schobeß A,
Müller W, Baethmann M, Leiz S, Gehrmann A, Makowski C, Fiedler
B, Böswald M, Weisbrod T, Kintzel K, Franz C, Feickert HJ, Kühl
PG, Schneider M, Raab K, Beyer P, Kauther KD, Reiter HL, Heubner
G, Behl ES, Trefz FK, Hoffmann HG, Buss M, Olbertz DM, Hempel L,
Agricola G, Horneff G, Augustin KS, Mihatsch W, Kurre A, Colling
S, Burghard R, Soditt V, Feierfeil K, Hornbrook D, Kellner L,
Pernice W, Schirmer D, Alber M, Geerken S, Kratz M, Köhler A,
Knuf M, Repinska T, Buller M, Becker JC, Niesytto C, Skopnik H,
Borusiak P, Verbeek T, Gabler I, Winkelmann T.
Author information
1
Division of Safety of Medicinal Products and Medical Devices,
Paul-Ehrlich-Institut, Federal Institute for Vaccines and
Biomedicines, Langen, Germany.
Abstract
PURPOSE:
A prospective, epidemiologic study was conducted to assess
whether the 2009 pandemic influenza A(H1N1) vaccination in
Germany almost exclusively using an AS03-adjuvanted vaccine (Pandemrix)
impacts the risk of Guillain-Barré syndrome (GBS) and its
variant Fisher syndrome (FS).
METHODS:
Potential cases of GBS/FS were reported by 351 participating
hospitals throughout Germany. The self-controlled case series
methodology was applied to all GBS/FS cases fulfilling the
Brighton Collaboration (BC) case definition (levels 1-3 of
diagnostic certainty) with symptom onset between 1 November 2009
and 30 September 2010 reported until end of December 2010.
RESULTS:
Out of 676 GBS/FS reports, in 30 cases, GBS/FS (BC levels 1-3)
occurred within 150 days following influenza A(H1N1)
vaccination. The relative incidence of GBS/FS within the primary
risk period (days 5-42 post-vaccination) compared with the
control period (days 43-150 post-vaccination) was 4.65 (95%CI
[2.17, 9.98]). Similar results were found when stratifying for
infections within 3 weeks prior to onset of GBS/FS and when
excluding cases with additional seasonal influenza vaccination.
The overall result of temporally adjusted analyses supported the
primary finding of an increased relative incidence of GBS/FS
following influenza A(H1N1) vaccination.
CONCLUSIONS:
The results indicate an increased risk of GBS/FS in temporal
association with pandemic influenza A(H1N1) vaccination in
Germany.
===================================================================
2.) Clinical Features of Post-Vaccination Guillain-Barré
Syndrome (GBS) in Korea.
==================================================================
J Korean Med Sci. 2017 Jul;32(7):1154-1159. doi:
10.3346/jkms.2017.32.7.1154.
Park YS1, Lee KJ2, Kim SW2, Kim KM2, Suh BC3.
Author information
1
Department of Neurology, Seoul National University Bundang
Hospital, Seoul National University College of Medicine,
Seongnam, Korea.
2
Division of Epidemic Intelligence Service, Korea Centers for
Disease Control and Prevention, Cheongju, Korea.
3
Department of Neurology, Kangbuk Samsung Hospital, Sungkyunkwan
University School of Medicine, Seoul, Korea. [email protected].
Abstract
Guillain-Barré syndrome (GBS) is the most common
immune-mediated polyradiculoneuropathy and it is also the most
commonly reported severe adverse event following immunization in
adults. To evaluate the results of clinical and laboratory
features of GBS after vaccination in Korea, we analyzed the
claims-based data from 2002 to 2014 using materials collected
for the Advisory Committee Vaccination Injury Compensation (ACVIC)
meeting including, clinical features, nerve conduction studies (NCSs),
cerebrospinal fluid (CSF) profiles, treatment, and outcomes.
Forty-eight compensated GBS cases (median age, 15 years;
interquartile range [IQR], 13-51; male:female ratio, 1:1) of 68
suspected GBS were found following immunization and all of them
with influenza immunizations with either monovalent (n = 35) or
trivalent (n = 13). Among them, 30 cases fulfilled the Brighton
criteria level 1-3 (62.5%). The median duration between the
onset of symptoms to nadir, duration of the nadir, and total
admission period were 3 (IQR, 2-7 days), 2 (IQR, 1-5 days), and
14 (IQR, 6-33 days) days, respectively. The most frequently
reported symptom was quadriparesis which was present in 36 cases
(75%) at nadir. CSF examination revealed albuminocytologic
dissociation in 25.0% and NCS was abnormal in 61.8%. After
treatment, most of them showed improvement. Clinical features
were similar to typical post-infectious GBS and there were both
demyelinating and axonal forms suggesting heterogeneous
pathogenic mechanism. In order to improve the diagnostic
certainty of post-vaccination GBS, careful documentation of
clinical features and timely diagnostic work-up with follow-up
studies are needed.
==========================================================================
3.) Lichen planus secondary to rabies vaccination.
==========================================================================
Dermatol Online J. 2017 Mar 15;23(3). pii: 13030/qt3hr3t4hs.
An I1, Demir V, İbiloğlu İ, Akdeniz S.
Author information
1
Department of Dermatology, Dicle University Faculty of
Medicine, Diyarbakır, Turkey. [email protected].
Abstract
Lichen planus (LP) is a papulosquamous disease withdistinctive
clinical manifestations. The etiology of LPremains unknown.
Recently, numerous cases of LPdeveloping after hepatitis B,
influenza, and combined DTaP-IPV-MMR vaccine have been
described. In thisreport, we present the second case of LP after
rabiesvaccination.
==========================================================================
4.) Lichen planus occurring after influenza vaccination: report
of three cases and review of the literature.
==========================================================================
Dermatology. 2010;221(4):296-9. doi: 10.1159/000321191. Epub
2010 Nov 3.
Sato NA1, Kano Y, Shiohara T.
Author information
1
Department of Dermatology, Kyorin University School of
Medicine, Tokyo, Japan. [email protected]
Abstract
Although influenza vaccine is thought to be effective and safe,
it occasionally causes systemic reactions such as toxic
epidermal necrolysis, bullous pemphigoid, lichen planus (LP),
etc. The period of increased risk of developing these events was
different depending on the immune responses induced by the
vaccination. We report 3 cases of LP which appeared after an
influenza vaccination. Our cases indicate that the period of
increased risk of developing vaccine-related LP was concentrated
within 2 weeks after vaccination, and that the vaccine alone
represents a triggering factor necessary for immune alteration
sufficient for the development of LP. Because these adverse
events tend to develop over a predictable time course, the time
of onset may give an important clue to the diagnosis of
vaccine-related diseases. We suggest that a history of recent
vaccination should be sought in all patients presenting with
linear
LP.==========================================================================
5.) Lichen planus after HBV vaccination in a child: a case
report from Nepal.
==========================================================================
J Dermatol. 2000 Sep;27(9):618-20.
Agrawal S1, Garg VK, Joshi A, Agarwalla A, Sah SP.
Author information
1
Department of Dermatology and Venereology, B.P. Koirala
Institute of Health Sciences, Dharan, Nepal.
Abstract
Vaccination against hepatitis B virus has rarely been
associated with lichen planus. We report a case of this kind in
a child from Nepal. A 12-year-old boy had developed generalized
itchy violaceous papules and plaques six weeks after the second
dose of hepatitis B virus vaccine. Serum HBsAg and HBeAb were
negative, but HBsAb was positive. New crops of generalized,
similar eruptions developed after the booster dose of vaccine.
All the lesions resolved within three months of systemic steroid
therapy. There was no recurrence after one year of follow up.
Awareness of such an association is necessary, especially in
children, because vaccination campaigns are increasing.
==========================================================================
6.) Lichen planus induced by hepatitis B vaccination: a new
case and review of the literature.
==========================================================================
Int J Dermatol. 2004 Aug;43(8):562-4.
Calista D1, Morri M.
Author information
1
Department of Dermatology, M. Bufalini Hospital, Cesena, Italy.
[email protected]
Abstract
In May 1996, as part of his routine antihepatitis B (hepB)
vaccination plan, a 28-year-old HbsAg-negative man, hospital
worker, received his first dose (20 microg) of a recombinant
vaccine (EngerixB-B, Smith Kline and Beecham, Belgium),
administered via deltoid injection. The patient was otherwise
healthy and taking no medication. Thirty days after the 2nd
booster dose, several pruritic, polygonal, purple, papules
appeared on the volar aspect of the patient's wrists. New
lesions gradually spread to the arms and trunk (Fig. 1). The
clinical diagnosis of lichen planus (LP) was confirmed by
histology, which revealed hyperorthokeratosis, hypergranulosis,
vacuolar degeneration of the basal layer cells and a dense,
band-like lymphocytic infiltrate in the superficial dermis. The
disease started to heal after treatment with topical clobetasol
propionate 0.05% and sun exposure during the following summer.
Five days after the 3rd booster dose, in November 1996, the
dermatosis relapsed on the forearms, trunk, and legs. On that
occasion, routine laboratory tests, including a complete blood
count, blood chemistry and liver function tests, were within
normal limits. Screening serologic tests for autoantibodies
including antinuclear antibodies, antidouble-stranded DNA,
anti-SS-A, anti-SS-B and anti-Sm were all negative. As a result
of the inadequate levels of antihepatitis B antibodies, less
than 10 IU/l in May 1998, in a high-risk patient who was
frequently exposed to blood and its products, an additional
booster dose was performed. Three days later a new recurrence of
disseminated lichen planus occurred. The patient was
successfully treated with prednisone 1 mg/kg/day for 2 weeks.
There was no recurrence the following year.
==========================================================================
7.) Hepatitis B vaccination and associated oral manifestations:
a non-systematic review of literature and case reports.
=========================================================================
Ann Med Health Sci Res. 2014 Nov;4(6):829-36. doi:
10.4103/2141-9248.144870.
Tarakji B1, Ashok N1, Alakeel R2, Azzeghaibi S1, Umair A1,
Darwish S1, Mahmoud R3, Elkhatat E3.
Author information
1
Department of Oral Maxillofacial Sciences, Alfarabi College of
Dentistry and Nursing, Riyadh, Saudi Arabia.
2
Department of Clinical Laboratory Sciences, King Saud
University, Alfarabi College of Medicine, Riyadh, Saudi Arabia.
3
Department of Restorative Dentistry Sciences, Alfarabi College
of Dentistry and Nursing, Saudi Arabia.
Abstract
Hepatitis B vaccine has been administered in children and
adults routinely to reduce the incidence of the disease. Even
though, hepatitis B vaccine is considered as highly safe, some
adverse reactions have been reported. A literature search was
carried out in PubMed, accessed via the National Library of
Medicine PubMed interface, searching used the following
keywords: Hepatitis B vaccine and complications from 1980 to
2014. A total of 1147 articles were obtained out of which
articles, which discuss the complications occurring orally or
occurring elsewhere in the body, which have the potential to
manifest orally after hepatitis B vaccination were selected. A
total of 82 articles were identified which included 58 case
series or case reports, 15 review articles, 4 cross sectional
studies, 3 prospective cohort studies, one retrospective cohort
study and a case control study. After reviewing the literature,
we observed that complications seen after Hepatitis B
vaccination are sudden infant death syndrome, multiple
sclerosis, chronic fatigue syndrome, idiopathic thrombocytopenic
purpura, vasculititis optic neuritis, anaphylaxis, systemic
lupus erytymatosus, lichen planus and neuro-muscular disorder.
Of these complications, some are manifested orally or have the
potential to manifest orally. Although, most of the
complications are self-limiting, some are very serious
conditions, which require hospitalization with immediate medical
attention.
==========================================================================
8.) A case-series of adverse events, positive re-challenge of
symptoms, and events in identical twins following hepatitis B
vaccination: analysis of the Vaccine Adverse Event Reporting
System (VAERS) database and literature review.
==========================================================================
Clin Exp Rheumatol. 2004 Nov-Dec;22(6):749-55.
Geier MR1, Geier DA.
Author information
1
The Genetic Centers of America, MedCon, Inc., Silver Spring,
Maryland 20905, USA. [email protected]
Abstract
OBJECTIVES:
Adverse events and positive re-challenge of symptoms reported
in the scientific literature and to the Vaccine Adverse Event
Reporting System (VAERS) following hepatitis B vaccination (HBV)
were examined.
METHODS:
The VAERS and PubMed (1966-2003) were searched for autoimmune
conditions including arthritis, rheumatoid arthritis, myelitis,
optic neuritis, multiple sclerosis (MS), Guillain Barré Syndrome
(GBS), glomerulonephritis, pancytopenia/thrombocytopenia,
fatigue, and chronic fatigue, and Systemic Lupus Erythematous (SLE)
following HBV.
RESULTS:
HBV was associated with a number of serious conditions and
positive re-challenge or significant exacerbation of symptoms
following immunization. There were 415 arthritis, 166 rheumatoid
arthritis, 130 myelitis, 4 SLE, 100 optic neuritis, 101 GBS, 29
glomerulonephritis, 283 pancytopenia/thrombocytopenia, and 183
MS events reportedfollowing HBV A total of 465 positive
re-challenge adverse events were observed following adult HBV
that occurred sooner and with more severity than initial adverse
event reports. A case-report of arthritis occurring in identical
twins was also identified.
CONCLUSIONS:
Evidence from biological plausibility, case-reports,
case-series, epidemiological, and now for positive re-challenge
and exacerbation of symptoms, and events in identical twins was
presented. One would have to consider that there is causal
relationship between HBV and serious autoimmune disorders among
certain susceptible vaccine recipients in a defined temporal
period following immunization. In immunizing adults, the
patient, with the help of their physician, should make an
informed consent decision as to whether to be immunized or not,
weighing the small risks of the adverse effects of HBV with the
risk of exposure to deadly hepatitis B virus.
==========================================================================
9.) A one year followup of chronic arthritis following rubella
and hepatitis B vaccination based upon analysis of the Vaccine
Adverse Events Reporting System (VAERS) database.
==========================================================================
Clin Exp Rheumatol. 2002 Nov-Dec;20(6):767-71.
Geier DA1, Geier MR.
Author information
1
MedCon, Inc., Silver Spring, Maryland, USA. [email protected]
Abstract
OBJECTIVES:
This analysis examined the incidence rate of chronic arthritis
adverse reactions reported following adult rubella and hepatitis
B vaccinations. In this analysis, etiologic mechanisms for
chronic arthritis following adult rubella and hepatitis B
vaccines were also explored.
METHODS:
The Vaccine Adverse Events Reporting System (VAERS) database
was analyzed for the incidence rate of reported cases of chronic
arthritis in comparison to Tetanus-diphtheria (Td) and tetanus
toxoid adult vaccine control groups.
RESULTS:
Chronic arthritis adverse reactions following adult rubella
vaccination were primarily reported in females (female/male
ratio = 3.0), at about 45 years-old, and at a mean onset time of
10-11 days following vaccination. Chronic arthritis adverse
reactions following adult hepatitis B vaccination were also
primarily reported in females(female/male ratio = 3.5), at about
33 years-old, and with a mean onset time of 16 days following
vaccination. The incidence rates of chronic arthritis following
adult rubella and adult hepatitis B vaccinations were
statistically significantly increased, by chi 2 analysis, in
comparison to the adult vaccine control groups. The attributable
risk of chronic arthritis following adult rubella vaccine ranged
from 32 to 53 and from 5.1 to 9.0 following adult hepatitis B
vaccine in comparison to the adult vaccine control groups.
CONCLUSION:
This study revealed that adult rubella and adult hepatitis B
vaccines were statistically associated with chronic arthritis
which persisted for at least one year. The etiology for these
adverse reactions may involve autoimmune mechanisms.
Furthermore, potential biases in the reporting rates of adverse
reactions to VAERS were not observed.
=========================================================================
10.) Hepatitis B vaccination and adult associated
gastrointestinal reactions: a follow-up analysis.
=========================================================================
Hepatogastroenterology. 2002 Nov-Dec;49(48):1571-5.
Geier DA1, Geier MR.
Author information
1
Genetic Centers of America, Silver Spring, MD, USA.
Abstract
BACKGROUND/AIMS:
Hepatitis B is the most important infectious cause of acute and
chronic liver disease. Hepatitis B vaccine, a highly purified,
genetically engineered, single antigen vaccine, has generally
been accepted as a safe vaccine. In 2000, the Institute of
Medicine noted that few vaccines for any disease have been
actively monitored for adverse effects over long periods and
encouraged evaluation of active long-term monitoring studies of
large populations to further evaluate the relative safety of
vaccines. The aim of this study was to accept the charge of the
2000 Institute of Medicine Report and extend our own work to
determine the frequency of gastrointestinal adverse reactions
after hepatitis B vaccination and determine if this frequency
was increased over the background rate of gastrointestinal
conditions in the U.S. adult population.
METHODOLOGY:
A retrospective examination of the Vaccine Adverse Events
Reporting System (VAERS) database from July 1990 through August
1999 for hepatitis B vaccination and associated gastrointestinal
reactions was made. Additionally, as controls, hepatitis A and
rubella vaccination associated gastrointestinal adverse
reactions reported to the Vaccine Adverse Events Reporting
System in adults were analyzed.
RESULTS:
Our analysis shows that the 40-year-old female population
between four to eight days after hepatitis B vaccination was at
increased risk for developing gastrointestinal reactions.
CONCLUSIONS:
Hepatitis B vaccination was statistically associated by chi 2
analysis with gastrointestinal reactions including: hepatitis,
gastrointestinal disease and liver function test abnormalities
in comparison to our vaccine control groups. The reaction rate
observed is outweighed by the benefits of the vaccine. Further
analysis is needed to determine the mechanisms by which
hepatitis B vaccine is associated with gastrointestinal
reactions.
=========================================================================
11.) Allergic reactions to Japanese encephalitis vaccine.
=========================================================================
Immunol Allergy Clin North Am. 2003 Nov;23(4):665-97.
Plesner AM1.
Author information
1
Department of Medical Officers of Health, Copenhagen County,
Islands Brygge 67 DK-2300 Copenhagen S, Denmark. [email protected]
Erratum in
Immunol Allergy Clin North Am. 2004 May;24(2):335.
Abstract
The JEV widely is used in Asian countries each year and is an
important vaccine for travelers to the East from other parts of
the world. JE virus is a zoonotic disease with natural
reservoirs and cannot be eliminated. Although a declining
incidence of JE has been observed in Asia because of reduced
transmission by agricultural approaches and vaccination, the
most important control measure now, and in the future, is
vaccination of humans against JE. The inactivated vaccine,
produced from infected mouse-brain-derived tissue, is the only
commercially available vaccine. There are several concerns with
the use of this vaccine. It is expensive, requires two or three
doses to achieve protective efficacy, and, in practice, requires
further booster doses to maintain immunity. The apparent
increase in allergic reactions in the first part of the 1990s
has set focus on the safety of the JEV. A cheap, live attenuated
SA 14-14-2 vaccine is used almost exclusively in China and parts
of Korea, but there have been no trials of SA 14-14-2 vaccine
outside JE endemic countries. The vaccine seems to be highly
efficient, and few adverse events have been observed; however,
PHK cells are used for the production of this vaccine, and these
cells are not approved by the WHO. A satisfactory cell substrate
is needed. A committee under the WHO has proposed that for the
live JEV, there should be validity of the assays for retrovirus
when applied to PHK cell substrate and validity of the mouse
assays for neurovirulence. Further information should be
reviewed on the long-term follow-up of recipients of the
vaccine. Several new types of vaccines have reached the phase of
clinical trials; however, studies remain to be completed. Until
a new vaccine is available, the priority of surveillance of
adverse events and the continuous reporting of such events to
the users of the vaccines must be of importance. This fact is
highlighted by the possibility of the varying frequency of
adverse events with different batches over the years. The WHO
offers information and recommendations for vaccines in the EPI
and issues a series of updated papers on other vaccines that are
of international public health importance (eg, JEV). The
development of alternative efficient, safe, and appropriately
priced JEVs is recommended, as is intensified surveillance of
adverse events. Prospective vaccine studies of safety may be
limited because of sample size and because rare adverse events
may not be detected. Several new initiatives have been taken to
improve surveillance of adverse events to vaccines within the
past 10 years. In Japan, there is an increasing awareness of the
importance of efforts taken to improve vaccine safety, and
surveillance of adverse events and possibilities of compensation
for vaccine-related injuries are in place. In Vietnam, a
database to detect adverse events after vaccination has been
established; the project involves active visits to data
collectors at the vaccination sites. Comparative studies of
adverse events, such as one recent study from Japan and the
United States, are important for the evaluation of the reporting
systems. The reporting rate for JEV adverse events from Japan
was approximately one order of magnitude lower than that in the
United States. Japan had strict predefined reporting criteria
and time limits for observations. If time limits for the
observation are too strict (eg, defining a possible neurologic
reaction to occur within 1 week after vaccination), later
reactions will not be included (eg, if ADEM is elicited by a
vaccine, the symptoms cannot be expected to occur until weeks
after the vaccination). The passive surveillance systems have
limitations with an underreporting of adverse events, depending
on clinical seriousness, temporal proximity to vaccination,
awareness of healthcare workers, and tradition of reporting
particular events. In developed countries, surveillance of
adverse events is formalized, although not necessarily optimal.
An increase in reporting would be expected when the reporting of
adverse events is mandatory. Reports have been sent to VAERS,
the Vaccine Safety Datalink Project, and the European Union
Pharmacovigilance System. A Brighton collaboration has been
implemented to enhance comparability of vaccine safety data.
Public health authorities in specific countries, such as the CDC
in the United States and the National Advisory Committee in
Canada, regularly have published information on the JE situation
in Asia and the preventive measures to be taken, including
information on the vaccines and adverse reactions. The
conventional recommendation is that travelers should be
vaccinated if they will spend more than 1 month in a JE endemic
area or in areas with epidemic transmission with even shorter
periods. Although the risk for JE for short-term travelers is
considered small (1 case per 1 million travelers per year),
sporadic cases, including deaths, have been reported among
tourists traveling to endemic areas. Risk for travelers in rural
districts in the season of risk is considerably higher (range, 1
case per 5000 travelers to 1 case per 20,000 travelers per
week). Doctors who advise travelers should be updated on the
latest JE occurrences in Asia. Updates on the JE situation can
be found on bulletins at http://www.promedmail.org or are
available from the WHO or CDC. The allergic reactions primarily
described after vaccination with the inactivated
mouse-brain-derived JEV have been observed in several countries
during the 1900s. Allergic reactions, including the
mucocutaneous and neurologic reactions reported after JE
vaccination, may vary in frequency, and these reactions should
be evaluated meticulously yearly. This step enables
recommendations, including information on possible side effects,
to be given in an optimal way.
=========================================================================
12.) The web and public confidence in MMR vaccination in Italy.
========================================================================
Vaccine. 2017 Aug 16;35(35 Pt B):4494-4498. doi:
10.1016/j.vaccine.2017.07.029. Epub 2017 Jul 20.
Aquino F1, Donzelli G2, De Franco E3, Privitera G1, Lopalco
PL4, Carducci A2.
Author information
1
Department of Translational Research, N.T.M.S. - University of
Pisa, Via Savi 10, 56126 Pisa, Italy.
2
Department of Biology - University of Pisa, Via S. Zeno 35,
56127 Pisa, Italy.
3
Division of Public Health and Nutrition - Area of Pisa, Azienda
USL Toscana Nord Ovest, Galleria Gerace 14, 56124 Pisa, Italy.
4
Department of Translational Research, N.T.M.S. - University of
Pisa, Via Savi 10, 56126 Pisa, Italy. Electronic address:
[email protected].
Abstract
Measles, mumps and rubella (MMR) vaccination coverage in Italy
has been decreasing starting from 2012 and, at the present, none
of the Italian regions has achieved the goal of 95% coverage
target. A decision of the Court of Justice of Rimini in March
2012 that awarded vaccine-injury compensation for a case of
autism has been indicated as a probable trigger event leading to
a reduction of vaccine confidence in Italy. The aim of the study
was to explore the relationship between MMR vaccination coverage
to online search trends and social network activity on the topic
"autism and MMR vaccine", during the period 2010-2015. A
significant inverse correlation was found between MMR
vaccination coverage and Internet search activity, tweets and
Facebook posts. New media might have played a role in spreading
misinformation. Media monitoring could be useful to assess the
level of vaccine hesitancy and to plan and target effective
information campaigns.
=========================================================================
13.) Media coverage of the measles-mumps-rubella vaccine and
autism controversy and its relationship to MMR immunization
rates in the United States.
=========================================================================
Pediatrics. 2008 Apr;121(4):e836-43. doi:
10.1542/peds.2007-1760.
Smith MJ1, Ellenberg SS, Bell LM, Rubin DM.
Author information
1
Division of Infectious Diseases, Children's Hospital of
Philadelphia, Philadelphia, Pennsylvania, USA. [email protected]
Abstract
OBJECTIVE:
The purpose of this work was to assess the association between
media coverage of the MMR-autism controversy and MMR
immunization in the United States.
METHODS:
The public-use files of the National Immunization Survey were
used to estimate annual MMR coverage from 1995 to 2004. The
primary outcome was selective measles-mumps-rubella nonreceipt,
that is, those children who received all childhood immunizations
except MMR. Media coverage was measured by using LexisNexis, a
comprehensive database of national and local news media. Factors
associated with MMR nonreceipt were identified by using a
logistic regression model.
RESULTS:
Selective MMR nonreceipt, occurring in as few as 0.77% of
children in the 1995 cohort, rose to 2.1% in the 2000 National
Immunization Survey. Children included in the 2000 National
Immunization Survey were born when the putative link between MMR
and autism surfaced in the medical literature but before any
significant media attention occurred. Selective nonreceipt was
more prevalent in private practices and unrelated to family
characteristics. MMR nonreceipt returned to baseline before
sustained media coverage of the MMR-autism story began.
CONCLUSIONS:
There was a significant increase in selective MMR nonreceipt
that was temporally associated with the publication of the
original scientific literature, suggesting a link between MMR
and autism, which preceded media coverage of the MMR-autism
controversy. This finding suggests a limited influence of
mainstream media on MMR immunization in the United States.
=========================================================================
14.) Vaccine Adverse Events: Separating Myth from Reality.
=========================================================================
Spencer JP1, Trondsen Pawlowski RH1, Thomas S1.
Author information
1
Conemaugh Family Medicine Residency Program, Johnstown, PA,
USA.
Abstract
Vaccines are one of the most successful medical advances in
modern times. Most vaccine-preventable illnesses are unfamiliar
to modern parents. Because of this, parents are increasingly
questioning the necessity of immunizing their children,
especially because no vaccine is completely free of adverse
effects or the risk of complications. Family physicians should
be aware of the risks and benefits of recommended immunizations.
Thimerosal is currently used only in multidose vials of
influenza vaccine, and exposure through vaccines is not
associated with adverse neurologic outcomes. The measles, mumps,
and rubella vaccine is not associated with autism. Vaccines are
associated with local reactions, such as pain and erythema. The
rotavirus vaccine minimally increases the rate of
intussusception, whereas other vaccines minimally increase the
risk of syncope. Although immunization with the human
papillomavirus vaccine is recommended for all boys and girls,
vaccination rates remain low. Physicians should guide parents to
credible resources if they are considering vaccine refusal. If a
recommended vaccine is refused, proper documentation is
essential. The Vaccine Adverse Event Reporting System and
National Vaccine Injury Compensation Program track adverse
events and allow compensation for documented harms from
vaccinations
=========================================================================
15.) Refusal to Vaccinate Child Gets Mom Jail Time: A Deeper
Analysis
=========================================================================
October 7th 2017 at 10:15 am
Written By: Jefferey Jaxen
Source:http://www.greenmedinfo.com/blog/refusal-vaccinate-child-gets-mom-jail-time-deeper-analysis
“I want to make it perfectly clear. We’re leaving here today.
Dad’s picking the child up and he’s going to be vaccinated
regardless of what Mom did or didn’t do.”
These were the words of Oakland County judge Karen McDonald
during the open minutes of the recent court room proceedings
that continue to grab international headlines. Metro Detroit’s
Rebecca Bredow, the Mom, now sits in an Oakland Country jail
with a criminal record forever attached to her name. Her
9-year-old son is now in temporary custody of his father who is
ordered by the court to bring the child up to date on the boy’s
vaccination status, which will be up to eight vaccines “…as
rapidly as medically necessary.”
Unfortunately in America, the end result of cases like Bredow’s
are becoming more and more common.
Some are saying Bredow refused to vaccinate her child and is
getting what she deserved but is it really that simple? The
mainstream, corporate media narrative is attempting to paint a
picture that Bredow’s case is an uncommon, one-and-done
occurrence. The narrative is also suggesting that the family
court process, when vaccination status is concerned, is a stone
solid justice machine based on 'settled vaccine science.' The
reality is that the judge and the court are taking a known and
dangerous medical risk with another person’s child that they
have no right to take. Do courts have the right to order an
unavoidably unsafe medical intervention like vaccination in
custody cases?
At minute 3:30 Judge McDonald makes clear her forced
vaccination agenda.
Joel Dorfman of Michigan for Vaccine Choice, a group that
advocates for parents’ rights to refuse vaccines told the
Detroit Free Press, “If this child is injured as a result of
being given eight immunizations, who do you think is going to
take care of the child? The judge?”
According to Judge McDonald, Bredow’s case is about her refusal
to follow court orders she previously agreed to. McDonald ruled
Bredow was in criminal contempt for not following a 2016
agreement to vaccinate her child. However Bredow says that her
attorney at the time signed the order and advised her not to
worry since she had filed state waivers and vaccine exemptions
each year in Michigan for her child. In Michigan, parents or
guardians of children enrolled in public and private schools are
required to attend an educational session before they are
granted waivers.
Charlie Langton ✔@charlielangton
Mom goes to jail for not vaccinating her kid. But what is this
case really about? On @FOX2News - @WWJ950
3:54 PM - Oct 4, 2017
2121 Replies
2020 Retweets
1515 likes
Twitter Ads info and privacy
Lecturing from the bench, Judge McDonald told Bredow “I
understand you love your children. But what I don’t think you
understand is that your son has two parents, and dad gets a
say,” Her statement seems reasonable yet it is important to note
that Bredow has primary caregiver status. Digging deeper into
the information of the case, Judge McDonald’s recent ruling
gives physical custody of the child to the ex-husband James
Horne. In the past, Child Protective Services did an
investigation on Horne and the case was confirmed as a Category
3 revealing a preponderance of evidence against him which the
court knew about.
What about medical expert testimony? Although Bredow’s case
didn’t involve the testimony of an expert witness or medical
professional, this tactic is often a nonstarter in US courts.
The courts don’t decide and rule on the science, their job is to
weigh the evidence. For each doctor or expert witness brave
enough to go on record against the safety of vaccines in a given
case, there are many more doctors who are will testify for them.
In addition, all US health agencies and organizations still toe
the line for the false ‘safe and effective’ vaccine narrative
and refuse to factor in any new or highly relevant information
that says otherwise.
During the recent ruling, Judge McDonald appeared to be reading
from a prewritten statement when handing down her decision
suggesting that she did not factor in the day’s testimony and
dialogue. If that is the case, perhaps McDonald’s prewritten
decision was in response to the attention Bredow drew to the
case by going to the media. Section 600.1715 of Michigan’s
Revised Judicature Act of 1961 states:
“If the contempt consists of the omission to perform some act
or duty that is still within the power of the person to perform,
the imprisonment shall be terminated when the person performs
the act or duty or no longer has the power to perform the act or
duty…”
The “act or duty” to vaccinate Bredow’s 9-year-old child was
ordered by the court to be done by the ex-husband. In addition,
Bredow no longer had the power to perform the act or duty in
question. It seems that, given the language of the act, Bredow’s
jail time was handed down as a warning and a lesson rather than
a necessary legal measure.
=======================================================================
16.) Why Japan banned MMR vaccine
=======================================================================
by JENNY HOPE, Daily Mail
source:http://www.dailymail.co.uk/health/article-17509/Why-Japan-banned-MMR-vaccine.html
Japan stopped using the MMR vaccine seven years ago - virtually
the only developed nation to turn its back on the jab.
Government health chiefs claim a four-year experiment with it
has had serious financial and human costs.
Of the 3,969 medical compensation claims relating to vaccines
in the last 30 years, a quarter had been made by those badly
affected by the combined measles, mumps and rubella vaccine,
they say.
The triple jab was banned in Japan in 1993 after 1.8 million
children had been given two types of MMR and a record number
developed non-viral meningitis and other adverse reactions.
Official figures show there were three deaths while eight
children were left with permanent handicaps ranging from damaged
hearing and blindness to loss of control of limbs.
The government reconsidered using MMR in 1999 but decided it
was safer to keep the ban and continue using individual vaccines
for measles, mumps and rubella.
The British Department of Health said Japan had used a type of
MMR which included a strain of mumps vaccine that had particular
problems and was discontinued in the UK because of safety
concerns.
The Japanese government realised there was a problem with MMR
soon after its introduction in April 1989 when vaccination was
compulsory. Parents who refused had to pay a small fine.
An analysis of vaccinations over a three-month period showed
one in every 900 children was experiencing problems. This was
over 2,000 times higher than the expected rate of one child in
every 100,000 to 200,000.
The ministry switched to another MMR vaccine in October 1991
but the incidence was still high with one in 1,755 children
affected. No separate record has been kept of claims involving
autism.
Tests on the spinal fluid of 125 children affected were carried
out to see if the vaccine had got into the children's nervous
systems. They found one confirmed case and two further suspected
cases.
In 1993, after a public outcry fuelled by worries over the flu
vaccine, the government dropped the requirement for children to
be vaccinated against measles or rubella.
Dr Hiroki Nakatani, director of the Infectious Disease Division
at Japan's Ministry of Health and Welfare said that giving
individual vaccines cost twice as much as MMR 'but we believe it
is worth it'.
In some areas parents have to pay, while in others health
authorities foot the bill.
However, he admitted the MMR scare has left its mark. With
vaccination rates low, there have been measles outbreaks which
have claimed 94 lives in the last five years.
Follow us: @MailOnline on Twitter | DailyMail on Facebook
=======================================================================
17.) Japanese Government Continues to Ban the MMR Vaccine
========================================================================
source:https://vactruth.com/2016/06/23/japanese-government-bans-mmr-vaccine/
For many years, controversy has surrounded the three-in-one
vaccine against measles, mumps, and rubella. Most notably, the
MMR vaccine is infamous for its disputed connection to autism,
and despite the fact that it has been blamed in vaccine courts
for causing autism, vaccine supporters still deny its fault in
skyrocketing rates of autism spectrum disorder, which is at
least one in 68 children, with even higher rates of diagnosis
among boys. [1, 2]
However, the vaccine has other serious risks in addition to the
relationship it has with unmanageable numbers of autism in
children, which has led to a ban of this vaccine in one
industrialized nation.
The Japanese government banned the measles, mumps, and rubella
vaccine from its vaccination program in 1993, after a record
number of children developed adverse reactions, including
meningitis, loss of limbs, and death. [3]
The MMR Vaccine’s Tragic History in Japan
The MMR vaccine was introduced in Japan in April 1989, and
parents who refused the compulsory vaccine were fined. After
three months of analysis, officials realized that one in 900
children developed adverse reactions to the vaccine, a rate that
was 2,000 times higher than the expected rate.
Officials had hoped to resolve the problem by switching to
another version of the vaccine, but the excessive amount of
adverse reactions persisted, with one in 1,755 children
affected. Testing of 125 children’s spinal fluid determined that
the vaccines had entered one child’s nervous system, with two
additional suspected cases.
Four years later, in 1993, the government removed the MMR
mandate against measles and rubella. A doctor from Japan’s
Ministry of Health and Welfare admitted that the separate,
individual doses of measles and rubella cost twice as much to
administer, and he defended the decision, stating, “but we
believe it is worth it.” Furthermore, a member of the health
ministry also stated that the ban has not caused an increase in
deaths from measles. [4]
Japanese officials were also concerned about the MMR vaccine
causing additional cases of mumps, citing numerous studies in
The Lancet. [5]
Mumps and hepatitis B vaccines are not part of the national
immunization program in Japan. [6]
What Many Parents Don’t Know About the MMR Vaccine
The list of adverse reactions to the MMR vaccine, straight from
Merck’s vaccine package inserts, is long and alarming. A
shortened version of the vaccine damage associated with the MMR
vaccine includes vomiting, diarrhea, anaphylaxis, ear pain,
nerve deafness, diabetes, arthritis, myalgia, encephalitis,
febrile seizures, pneumonia, and death. [7, 8]
A search of the Vaccine Adverse Event Reporting System (VAERS)
database shows the following statistics from the United States:
over 75,000 adverse events have been reported from any
combination of measles, mumps, and rubella vaccines, including,
most notably:
78 deaths
85 cases of deafness
48 cases of decreased eye contact
92 cases of developmental delay
855 reported cases of autism
116 cases of intellectual disability
401 reports of speech disorders
276 reports of loss of consciousness
143 cases of encephalitis
74 cases of meningitis
111 cases of Guillain-Barré syndrome
692 cases of gait disturbance (not being able to walk normally)
748 cases of hypokinesia (partial or complete loss of muscle
movement)
653 reports of hypotonia (poor muscle tone)
4874 reports of seizures, including febrile convulsions and
tonic clonic seizures
1576 cases of cellulitis (a potentially serious skin infection)
And finally, in some cases, the vaccine has caused the very
diseases it is supposed to prevent, with the following data
reported to VAERS:
147 cases of measles
384 cases of mumps
29 cases of rubella [9]
The number of adverse events following vaccination are vastly
underreported, as acknowledged by the Centers for Disease
Control and Prevention (CDC). The National Vaccine Information
Center estimates that less than one to ten percent of adverse
reactions to vaccines are reported. Many of the numbers reported
above could therefore be multiplied by one hundred to determine
a more accurate amount of adverse reactions. [10, 11]
Japan Takes a Protective Stance Against Other Vaccines, Too
The flu vaccine has also been the subject of controversy in
Japan, after 100 deaths occurred from the vaccine by the end of
2009. Japan’s health ministry has been criticized for for its
cautious stance against vaccines, but so far, government
officials have wisely defended their position, citing public
safety as the paramount concern.
Finally, the Japanese government has also taken a protective
stance against vaccines on behalf of its young girls, suspending
the human papilloma virus (HPV) vaccine in 2013 after numerous
cases of serious adverse events were reported, with one report
citing as many as 1,968 adverse events, 358 of which were
classified as serious.
Japanese officials were concerned about the well-being of their
young citizens, despite having invested $187 million in the
program. Damage payments to only a fraction of the victims who
have suffered adverse reactions to the HPV vaccine have reached
$6 million. [12]
Additionally, since 2011, at least 38 infants have been
reported to have died after they had been vaccinated against
haemophilus influenza B and streptococcus pneumonia, according
to records compiled by the health ministry in Japan.
Japanese Officials Speak Out
Japan has been criticized for being behind the times when it
comes to vaccination. Vaccine advocates claim that Japan has not
kept pace with other developed countries regarding the use of
vaccines. Despite listing 110 infectious diseases in a
government registry, Japan offers vaccines for only 22 of those.
Some Japanese health experts disagree, however. Hiroko Mori, a
vaccine researcher, is one of those experts. He was the former
head of the infectious disease division at Japan’s National
Institute of Public Health.
He has noted that Japan has one of the lowest infant mortality
rates in the world and has advocated for fewer vaccines, stating
that the country’s excellent sanitation and nutrition has
boosted children’s health.
He observed,
“Medicine is supposed to be about healing, but babies who
cannot speak are being given unnecessary shots because parents
are scared. Children are losing their ability to heal naturally.
There are so many people who have suffered side effects. All we
are asking is to establish the right to say ‘no.’ The right to
choose should be recognized as a fundamental human right.”
Tetsuo Nakayama, Dean of Kitasato University’s Graduate School
of Infection Control Sciences, is an expert who supports
vaccines, but he, too, acknowledges the risks of vaccination,
stating that:
“There is no guarantee that your child will not be that one out
of 1,000. You have to compare the risks between the side effects
and what will happen if you are infected with the disease
naturally.
Under the existing law, the decision to vaccinate your child or
not is basically left up to the parents, but there is not enough
information out there for them to make an informed decision.”
Masako Koga, a former representative of the Consumers Union of
Japan, has shared his concerns about the ulterior motives behind
mass vaccination programs:
“Vaccines should only be given to those who need them but that
is not happening. The global industry is being driven by a
strategy that promotes VPD [vaccine preventable diseases].
We must put a stop to it. Vaccines have close ties to money.
From development to circulation to research on side effects,
there are a lot of vested interests involved.”
He also summarized what motivates many parents’ decisions not
to vaccinate their children:
“There is no knowing who will suffer side effects as a result
of vaccination.
[Proponents of vaccination] say the chance of suffering a side
effect is 1 in a million. For parents, however, that one is
everything.”
Conclusion
Japanese officials have made decisions that value the health
and safety of their citizens when they have removed vaccines
with dangerous side effects from their national vaccination
program.
Japan boasts a low infant mortality rate, despite — or perhaps
because of — mandating only a fraction of the vaccines required
by other developed countries, including the United States.
If you wish to learn more about the harmful ingredients in
vaccines or the potential adverse reactions, we have compiled an
easy-to-navigate list of vaccine package inserts from the
manufacturers that you can view or download here.
Has your child suffered an adverse reaction to the MMR vaccine
or the HPV vaccine, both of which have been removed from Japan’s
national vaccination program? If so, please share your story in
our comment section below.
=======================================================================
18.) A mass sterilization exercise’: Kenyan doctors find
anti-fertility agent in UN tetanus vaccine
======================================================================
Thu Nov 6, 2014 - 2:29 pm EST
Autthor: Steve Weatherbe
Source:https://www.lifesitenews.com/news/a-mass-sterilization-exercise-kenyan-doctors-find-anti-fertility-agent-in-u
UPDATE (Nov. 12): Kenya's government has launched an
investigation into the Catholic Church's allegations. See follow
up article here.
Kenya’s Catholic bishops are charging two United Nations
organizations with sterilizing millions of girls and women under
cover of an anti-tetanus inoculation program sponsored by the
Kenyan government.
According to a statement released Tuesday by the Kenya Catholic
Doctors Association, the organization has found an antigen that
causes miscarriages in a vaccine being administered to 2.3
million girls and women by the World Health Organization and
UNICEF. Priests throughout Kenya reportedly are advising their
congregations to refuse the vaccine.
“We sent six samples from around Kenya to laboratories in South
Africa. They tested positive for the HCG antigen,” Dr. Muhame
Ngare of the Mercy Medical Centre in Nairobi told LifeSiteNews.
“They were all laced with HCG.”
Dr. Ngare, spokesman for the Kenya Catholic Doctors
Association, stated in a bulletin released November 4, “This
proved right our worst fears; that this WHO campaign is not
about eradicating neonatal tetanus but a well-coordinated
forceful population control mass sterilization exercise using a
proven fertility regulating vaccine. This evidence was presented
to the Ministry of Health before the third round of immunization
but was ignored.”
But the government says the vaccine is safe. Health Minister
James Macharia even told the BBC, “I would recommend my own
daughter and wife to take it because I entirely 100% agree with
it and have confidence it has no adverse health effects.”
And Dr. Collins Tabu, head of the Health Ministry’s
immunization branch, told the Kenyan Nation, that “there is no
other additive in the vaccine other than the tetanus antigen.”
Tabu said the same vaccine has been used for 30 years in Kenya.
Moreover, “there are women who were vaccinated in October 2013
and March this year who are expectant. Therefore we deny that
the vaccines are laced with contraceptives.”
Newspaper stories also report women getting pregnant after
being vaccinated.
Responds Dr. Ngare: “Either we are lying or the government is
lying. But ask yourself, ‘What reason do the Catholic doctors
have for lying?’” Dr. Ngare added: “The Catholic Church has been
here in Kenya providing health care and vaccinating for 100
years for longer than Kenya has existed as a country.”
Dr. Ngare told LifeSiteNews that several things alerted doctors
in the Church’s far-flung medical system of 54 hospitals, 83
health centres, and 17 medical and nursing schools to the
possibility the anti-tetanus campaign was secretly an
anti-fertility campaign.
Why, they ask does it involve an unprecedented five shots (or
“jabs” as they are known, in Kenya) over more than two years and
why is it applied only to women of child-bearing years, and why
is it not being conducted without the usual fanfare of
government publicity?
“Usually we give a series three shots over two to three years,
we give it anyone who comes into the clinic with an open wound,
men, women or children.” said Dr. Ngare. “If this is intended to
inoculate children in the womb, why give it to girls starting at
15 years? You cannot get married till you are 18.” The usual way
to vaccinate children is to wait till they are six weeks old.”
But it is the five-vaccination regime that is most alarming.
“The only time tetanus vaccine has been given in five doses is
when it is used as a carrier in fertility regulating vaccines
laced with the pregnancy hormone, Human Chorionic Gonadotropin (HCG)
developed by WHO in 1992.”
It is HCG that has been found in all six samples sent to the
University of Nairobi medical laboratory and another in South
Africa. The bishops and doctors warn that injecting women with
HCG , which mimics a natural hormone produced by pregnant women,
causes them to develop antibodies against it. When they do get
pregnant, and produce their own version of HCG, it triggers the
production of antibodies that cause a miscarriage.
“We knew that the last time this vaccination with five
injections has been used was in Mexico in 1993 and Nicaragua and
the Philippines in 1994,” said Dr. Ngare. “It didn’t cause
miscarriages till three years later,” which is why, he added,
the counterclaims that women who got the vaccination recently
and then got pregnant are meaningless.
Ngare said WHO tried to bring the same anti-fertility program
into Kenya in the 1990s. “We alerted the government and it
stopped the vaccination. But this time they haven’t done so.”
Ngare also contrasted the secrecy of this campaign with the
usual fanfare accompanying national vaccination efforts. “They
usually bring all the stakeholders together three months before
the campaign, like they did with polio a little while ago. And
they use staff in all the centres to give out the vaccine.” But
with this anti-tetanus campaign, “only a few operatives from the
government are allowed to give it out. They come with a police
escort. They take it away with them when they are finished. Why
not leave it with the local medical staff to administer?”
Brian Clowes of Human Life International in Virginia told
LifeSite News that WHO was not involved in the Nicaragua,
Mexican and Philippines campaigns. “They try to maintain a
spotless record. They let organizations like United Nations
Population Fund and USAID do the dirty work.”
In the previous cases, said Clowes, the vaccinators insisted
their product was pure until it was shown not to be. Then they
claimed the positive tests for HCG were isolated, accidental
contaminations in the manufacturing process.
LifeSiteNews has obtained a UN report on an August 1992 meeting
at its world headquarters in Geneva of 10 scientists from
“Australia, Europe, India and the U.S.A” and 10 “women’s health
advocates” from around the world, to discuss the use of
“fertility regulating vaccines.” It describes the “anti-Human
Chorionic Gonadotropin vaccine” as the most advanced.
One million Kenyan women and girls have been vaccinated so far
with another 1.3 million to go. The vaccination is targeting
women, according to the government, in order to inoculate their
children in the womb against tetanus as well. The government
says 550 children die of tetanus yearly.
In covering the contest of words the pro-government Nation
found plenty of women who had been vaccinated and were now
pregnant, even one who was the wife of a former Catholic priest
who left the Church to marry. The paper ignored Kenya’s reliance
on the Catholic medical system, while setting the bishops’ stand
in a questionable historical context of irrational responses
“largely based on religious beliefs,” the more recent murder of
vaccination teams in Nigeria, and even of CIA conspiracy
theories.
Why would the UN want to suppress the population in developing
countries? “Racism,” is Brian Clowes’ first explanation. “Also,
the developed countries want to get hold of their natural
resources. And lately, there is the whole bogus global warming
thing.”
Dr. Ngare said it was the Catholic Church’s hope that the
government could have resolved the matter quietly by testing the
vaccine. “But the government has chosen to be combative,”
forcing Kenya’s bishops and Catholic doctors to go public.
WHO’s Kenyan office and several WHO media contacts in
Washington, D.C. failed to respond to LifeSiteNews enquiries
over a 24-hour period.
=======================================================================
19.) Raila Odinga: Tetanus vaccination is a mass sterilization
on women
=======================================================================
Source:https://www.standardmedia.co.ke/article/2001254261/raila-odinga-tetanus-vaccination-is-a-mass-sterilization-on-women
Published Tue, September 12th 2017 at 00:00, Updated September
11th 2017 at 22:43 GMT +3
Opposition chief Raila Odinga has reignited the tetanus
vaccination debate, claiming it was a State-sponsored tool for
mass sterilisation. Raila, in rare support for the Catholic
Church which at some point opposed the vaccine, accused the
Government of what he termed 'State-sponsored infertility in
women'. In January 2014 and 2015, the Catholic bishops
vehemently opposed the vaccine, claiming that one-third of the
vials tested contained a hormone linked to birth control. The
church claimed that the vaccine was targeting women of
child-bearing age and that it contained elements that prevented
them from conceiving. Raila seems to be reading from a similar
script, claiming that multiple results from health experts had
confirmed that the tetanus vaccine used in the immunisation
exercise triggered infertility in women aged between 14 and 49.
Forced practice "Sterilisation without full, free, and informed
consent globally is an involuntary, coercive, and/or forced
practice and is a violation of fundamental human rights," said
Raila. The National Super Alliance (NASA) leader demanded that
the Government provide a complete list of all those who took
part in the vaccination, apologise to them, and explain how it
intends to reverse the damage. He said should NASA form the next
government, it will set up a task force to establish the
vaccine's victims, recommend appropriate compensation for them,
assign responsibility, and initiate ways of reversing the
damage. "This is the greatest crime against humanity ever
committed against the women of Kenya and the most diabolical
attempt at social engineering. No one can tell the worth of a
woman's fertility or sterility," said Raila. Growing populations
He said that as a result of the administration of the vaccine,
thousands of girls and women aged between 14 and 49 from the
fastest growing populations in the country will not have
children because of what he described as 'State-sponsored
sterilisation'. "Based on results that are now available to us,
it is clear that the tetanus vaccination is one of the most
callous human rights abuses committed against innocent girls and
women in Kenya," he said.
=======================================================================
20.) Infant Dies Following 5 Vaccine Doses
=======================================================================
Source:https://vactruth.com/2015/09/05/infant-dies-after-5-vaccine-doses/
Life after losing a loved one to vaccines is very painful. With
a heavy heart, we share Sebastian Ryan Morley’s story. He was a
healthy boy whose life ended after routine vaccinations.
Sebastian’s mother and grandmother have worked many years in
both the veterinary and human healthcare fields. What they were
taught in school led them to believe vaccines were safe, but now
they will never vaccinate again. We thank his family for coming
forward and sharing very important information the public isn’t
usually made aware of.
Sebastian’s grandmother, Valerie Murfin, shared:
“On December 11, 2002, when my grandson Sebastian was seven
months old, he was taken in for his six month well child
checkup. My daughter Natasha, who is his mother, was not bullied
into getting him vaccinated, she was just following what she
thought was good advice, what we both thought was good advice at
one time.
During this visit, Sebastian received the vaccines for DTaP,
hepatitis B, and HiB. This is five vaccine doses.
Sebastian began vomiting two days later and suffered jaundice.
After turning yellow around his mouth on 12-15-02, his mom took
him to the doctor because he became very lethargic. She was
begging for him not to die on the drive to the doctor’s office.
They admitted him to St. Peter Hospital, in Olympia, where he
stayed for two days.
Tests were run and it was found that Sebastian’s liver was
failing. After an ultrasound showed his liver stopped swelling,
they allowed him to be discharged, but warned my daughter not to
let him bump his head or anything, because he could bleed out.
Before being discharged, his doctor did more blood work and
told my daughter that she would call her, and that they assumed
Sebastian had hepatitis C somehow. This was six days after the
vaccines were given to him, on 12-17-02.”
Sebastian’s Brain Swelled and His Organs Shut Down
“That phone call came in on 12-20-15 and the news made my
daughter aware it was urgent. Sebastian was rushed to the ER at
Seattle Children’s Hospital. This is where he stayed and
suffered, for more than a month.
Towards the end of his stay, his eyes were no longer responsive
and one swelled up horribly; his choke reflex was gone. I think
his little brain had had enough of all the additional chemicals
he received while in the hospital. Sebastian’s brain had swelled
outside of his soft spot, they had no more hope for him, and
they wanted to turn off the machines.
My daughter came out of that room, looked at me and said,
‘Don’t let them kill my baby, Mom.’ I had to tell her he was
already gone. I watched the light die in my daughter’s eyes.
They did unplug him. He died in his mom and dad’s arms. He died
on January 22, 2003, at just 8 ½ months old. Forty-two days
after that well child visit, multiple organs of his shut down
and his little body couldn’t fight anymore.
My most helpless moment in my life was when I had seen the
ultrasound of Sebastian’s brain. By this time, all of his organs
were shutting down. They had him in a medically-induced coma for
about the last five days. The first three days, they would let
him wake up. The last two days, he stopped waking up, even when
coming out of the anesthesia.
Ultimately, if he hadn’t had the vaccines, none of this would
have happened.
My daughter did question the vaccines with the doctor that gave
them and her initial response was that they just never see that.
Although Sebastian’s doctor later on was more supportive of the
fact that vaccines could not be ruled out.
His cause of death listed on his death certificate is
‘Fulminant Liver Failure of Unknown Etiology,’ though the
pathologist stated clearly when he wrote, ‘Because this child
was vaccinated less than 24 hours prior to onset of illness, we
can NOT rule out the vaccines as causative.’
This is as close anyone gets to admission that the vaccines
were responsible.”
Vaccines Were The Only Plausible Cause Of Sebastian’s Liver
Failure
“In the days he was in the in the pediatric ICU at the
hospital, they did every test they could at the time. There was
no physiological or environmental reason for his liver failing.
I kept saying in the hospital, he was just vaccinated, to look
into the vaccines, but hospital staff did not make the
connection.
After Sebastian got sick and was taken to the hospital, that
was when my daughter also made the vaccine connection; I made
sure she was aware. Even the hospital doctors kept saying
vaccine reactions never happen. If I weren’t so vocal about it,
they wouldn’t have put the vaccines into the equation.
His pediatrician, who actually worked with him, has been very
helpful. She felt it was important enough to report Sebastian’s
reactions to the Vaccine Adverse Reaction Reporting System (VAERS).
We appreciated her acknowledging what happened to my grandson
but Sebastian’s passing has not stopped her from vaccinating
other children.
On Sebastian’s VAERS report, some things were reported
inaccurately. His age wasn’t listed correctly; he was seven
months old when he received the six month vaccines. We tried
fixing the discrepancies in the report, but we were not able to
get anyone who would correct them.
Considering most parents and doctors have never reported a
reaction to VAERS, due to not being informed and doctors not
being enforced to report reactions, mistakes can occur due to
being unfamiliar with the process and sometimes, the mistakes
occur on the VAERS end, because reps don’t list the information
provided correctly.
It is hard to read this. Knowing how Sebastian suffered from
the vaccines is why my family will not vaccinate further.”
VAERS
Sebastian Became More Sick After Each Hepatitis B Vaccine
“When I was in the hospital while Sebastian was there, I
overheard two nurses talking in the hall. One nurse was from the
pediatric ICU, where Sebastian was, and the other nurse was from
the neonatal unit, where the preemies are.
The nurse from the neonatal unit said to the other, ‘Well, we
just vaccinated all the babies, I hope we have beds open,’
meaning these premature babies that were doing fine in the
neonatal unit were just vaccinated and the neonatal nurse was
saying to the pediatric ICU nurse that they hope they have beds
open because these babies are now needing to be put on
respirators, since they are now in critical care. Because of the
hepatitis B vaccine just given to them, now the babies aren’t
doing well anymore. Doctors and nurses are more aware of vaccine
injuries than they lead you to believe.
My daughter is not able to be the champion for Sebastian as I
am. She is firmly against any vaccines, as you can imagine, but
in 2003, she was so vilified. She cheers me on in our fight. We
didn’t know about the National Vaccine Injury Compensation
Program until four years after Sebastian passed away. The
deadline to file was two years after he passed away, so it was
too late for us to file a claim.
My daughter found out she was pregnant the day after
Sebastian’s funeral. She had a baby girl who will be 12 in
September 2015. Sebastian’s sister has never been vaccinated and
is completely healthy.
Since this happened, my daughter did still see the same
pediatrician after having our granddaughter, but she never
pushed my daughter to vaccinate her. When my granddaughter was
about three years old, because of a change in insurance, this
forced my daughter to change pediatricians as well. She does
love and miss Dr. O’Leary and would probably still see her to
this day, if she had the choice, because she did not ever
pressure her to vaccinate again.
Even though she was the one who vaccinated Sebastian leading to
his passing, this doctor acknowledged his death and respected my
daughter’s choice as a mother to not vaccinate further. It was
important she had a doctor who understood why she no longer
wanted to vaccinate.
We believe it was the third hepatitis B vaccine that was the
cause of Sebastian’s health decline. After each set of hepatitis
B-containing vaccines, Sebastian got more and more sick. After
his first hep B shot was given two days after birth, he got
diarrhea and had a fever. He wouldn’t take the breast milk and
he got dehydrated. At three and four days old, my daughter was
devastated as a new mom who had to feed him formula the first
week.
After Sebastian received his second hepatitis B vaccine, along
with the other two month vaccines, his mom had gone to Six Flags
as a treat from her friends and Sebastian’s dad called her
saying he was very lethargic and had a fever. Sebastian’s dad
Jeff gave him medicine and watched him like a hawk, as he
seemingly got better.
Then his last set of shots given contained the hepatitis B
vaccine and his liver shut down.
The reason we also think the hep B vaccine played a big part in
this is because I can’t be vaccinated, because my immune system
mimics the disease when vaccinated to prevent it. I think
Sebastian may have gotten this trait from me. His immune system
saw hepatitis and killed his liver.
I have read studies since this happened, involving mice, which
show this can happen. I spent three years in allergy treatment
and through this process, my immunologist discovered my immune
system issues. After three years, he finally admitted there was
no way to fix my allergies.
In my family, I have a brother with Crohn’s disease and two
cousins with lupus; my mother has immune-mediated kidney
disease. I have an aunt with rheumatoid arthritis. All of us
were vaccinated in the past. Vaccines are clearly not great for
everybody. Physicians ask you about familial heart disease,
diabetes … why not immune system issues that may be
contraindicated for an immune system stimulant?”
Sebastian’s Story Has Never Made the News
“This is not the type of story any vaccine supporter, which
mainstream media serves, will allow out. My point is, until you
have a reaction close to you, until it hits you in the face, you
want to believe that those in power have your best interest at
heart.
If your doctor’s intake money is getting cut by the insurance
company for not following standardized prophylactic treatments,
and your sales rep is pressuring you into buying a cheap
vaccine, you buy in bulk and everyone gets vaccinated.
If you don’t vaccinate, your doctor’s bottom line is affected.
If your doctor fails to get a good family history, on any
disease in your family, and then pushes vaccines as 100 percent
safe and effective, then perhaps your doctor doesn’t have your
best interest in mind. This is a major income for all involved.”
Human Vaccines Are Just As Harmful As Pet Vaccines
“I worked in animal medicine for 26 years, I gave thousands of
vaccines to pets and I knew vaccines were causing many problems.
I had been gathering doubts about vaccines in that field for
years, which led me to leave the day practice I worked at and go
into a specialty field, where vaccines were not required.
At the vet clinic I worked for, I had serious issues with the
way Pfizer handled their sale; right away, they misrepresented
themselves, trying to sell us pet vaccines. In the 1990s, dogs
received what I call the alphabet shot (DHLPPC) for distemper,
hepatitis, leptospirosis, parainfluenza, parvovirus, and
coronavirus.
About 85-90 percent of smaller dogs under 20 pounds reacted to
the leptospirosis vaccine. These dogs were having many types of
reactions, some life-threatening, lifelong, and a few died
within hours. Bichon Frises, little white furry dogs, are
notorious for dropping dead after vaccination and a lot of
Bichon Frise owners and breeders won’t vaccinate their dogs.
So many dogs get the diseases they are vaccinated for. The fact
that so many dogs reacted was a clue. Back then, most people
still had bigger dogs. In the 1990s, there was a shift to the
smaller house dogs and that made the incidence of vaccine
reactions go up.
I asked the Pfizer rep, ‘Wouldn’t it be prudent to remove the
leptospirosis from the vaccine?’ He answered that it was not
cost effective to remove the ingredient. I had been gathering
doubts about vaccines in that field for a long time, but this
really opened my eyes.
After the Pfizer rep said it wasn’t cost effective to remove
leptospirosis, (which is stupid really, in my humble opinion,
because you just don’t put that ingredient in since it was
causing so much harm in these pets), I asked the rep what Pfizer
recommended. His answer was to give a high-dose steroid (their
high-priced product, another money maker), five minutes before
the vaccine.
They expected us to suppress the immune system with the steroid
Solu-Medrol®, to then immediately turn around and stimulate it
with the vaccine. That didn’t even make medical sense. As I said
before, that conversation was an eye opener. That guy must have
hated me because I began questioning every vaccine they rolled
out.
The rabies vaccine is the worst. It was causing many health
problems, including immune system problems and gut problems and
I witnessed some dogs would just drop dead on the table after
they received the rabies vaccine.
I also witnessed cats having a huge incidence of injection site
carcinomas. On the back of the shoulders where we injected them,
horrible tumors would develop. Even if surgery was performed,
ultimately this still killed the cats, a process I’ve seen take
two years. I brought this up to the vaccine rep because I was
concerned about what I was observing. The answer to that from
the rep was to vaccinate low on the leg, marking which vaccine
and which leg was injected. That way, if a tumor developed, you
can amputate.
At the time, the feline leukemia vaccine seemed to be the
culprit. I believe now that the vaccine manufacturers were aware
then that all of the vaccines could be problematic. In general,
any of the vaccines could cause this and they don’t test them
for causing cancer; now I see why, so they can’t be blamed when
they cause cancer.
If the companies are so into making money that they don’t care
about their patients, it doesn’t make sense to use their
products. Due to what I was learning and the vaccine reactions I
was witnessing, I left that practice to go into a specialty
field, where vaccines were not required. I have other stories,
but that was my light bulb moment. Regarding human vaccines
being safer, I just really believed that human medicine HAD to
be different and safer, but I was wrong.”
Our Family Will Never Vaccinate Again
“When I left, I started working at Washington State University
(WSU) in Pullman, WA, in their Veterinary Teaching Hospital’s
ICU, working with fourth year students on practical emergency
and critical care.This was years before Sebastian was even born.
It was a six hour drive from where my kids lived. I moved back
to Seattle after Sebastian died. I remained in specialty
medicine where I finished my career in internal medicine.
Ironically, a high percentage of our patients suffered from
vaccine-related immune-mediated disease.
My daughter also worked in the same veterinary clinic I did.
She stayed in the day practice a while longer and she was
working there when Sebastian was harmed. Sadly, I guess I didn’t
really convey the problems I saw in vaccines then. I really
believed that human medicine HAD to have more safety measures
than veterinary medicine.
I always wondered if I had yelled louder about the dangers of
vaccinating, would things be different? My daughter assures me
that she trusted her doctor and would have followed the
recommendations anyway. My family will never vaccinate anyone
under our care.
My daughter left the field of veterinary medicine, too. She now
works in the field of human medicine and is the medical
assistant for a doctor in a foot and ankle clinic. She also
doesn’t have to vaccinate. Since Sebastian passed away, my
daughter has always been up front with her employers, that she
won’t vaccinate patients and she discloses all information to
patients, if her practice gave vaccines.
Despite their differences of opinion, even with her knowledge
about the harm these vaccines are causing, she loves the doctor
she is working with, even though he is still a vaccine person,
though not as rigid as he once was. She calls this ‘little
steps,’ I think.
Prior to me becoming disabled after I broke my back in 2008, I
had subbed for a friend who went on vacation. At the one day
clinic I subbed in, I gave complete disclosure on vaccines,
letting them know I won’t give them. They didn’t invite me back.
I never went back to a day practice other than that.
Nowadays, my job is sharing Sebastian’s story, and working
against the mandates we know will be revisiting us in our state
of Washington. It takes a huge amount of my time. We have to
speak for those who can’t anymore. In this, we gather.
I am thankful to Sallie O. Elkordy, Host of ‘The Mary and
Sallie Show,’ who allowed me to share Sebastian’s story and to
let people know what is happening to children and pets after
vaccination.
Please, do your research, folks; know your risk of infection
for said disease, if you want to vaccinate. Our immune systems
work best when they are not chemically modified. I encourage
folks to share my grandson’s story. It is the only thing that
helps us come to terms with his death.
I gave Sebastian that first sink bath; it’s my favorite picture
of him. Oh, if I could, I would give him a head noogie, because
he would have just turned 13 this year and giving grandma a hug
just isn’t cool when you’re 13.
In all seriousness, we miss him so much. Not a day goes by that
he is not in our thoughts. The tears still fall. We would give
anything to see what his 13 year-old self would have been.
It was a very difficult thing for us to go through, losing him
the way we did. Like my daughter says, it’s been like a movie
that you cannot turn off or rewrite the script. The sequel, I
hope, sheds light on the other side, and stops the Vaccine
Holocaust. We had the bad luck to win the vaccine lottery; with
any decency in the country, countless others won’t be forced to.
If just one person chooses to inform themselves, if just one
more person wakes up, if one family is saved from this hole
carved out of our lives, then it gives us some peace. It gives
this little man a voice, when he had been silenced for so long.
We love you, Sebastian. You will not be forgotten.
Love, Your Grandma,
Valerie Murfin”
In Our Hearts He Will Be Remembered
His mother Natasha wrote:
“I miss my little monkey everyday. Some days are easier than
others but it’s still a very hard thing to swallow. My son’s
life was like a very short movie. And just like that, it was
over, with the saddest ending. I don’t want this to happen to
anyone else.”
=======================================================================
21.) NEW LYME VACCINE COMING SOON. CAVEAT EMPTOR––BUYER BEWARE!
Published on Published onAugust 4, 2017
=======================================================================
Author: Lori Dennis, MA,RP
Registered Psychotherapist, Speaker, Author of Lyme Madness
Source:https://www.linkedin.com/pulse/new-lyme-vaccine-coming-soon-caveat-emptorbuyer-lori-dennis-ma-rp/
In the Tech and Science section of Newsweek, July 25, 2017, the
headline reads: "Lyme disease vaccine on fast track for FDA
approval."http://www.newsweek.com/lyme-disease-vaccine-valneva-fda-approva-641796
Those of us in the Lyme world know that this is anything but
good news. In fact, for those of us ‘in the know’, it's
absolutely terrifying.
Why? you may ask. Why wouldn’t we want a vaccine to prevent
this illness? After all, we’ve been hearing just how
life-altering and debilitating this disease can be. Wouldn’t a
vaccine protect us from this potential fate?
I will give you five key reasons to consider why a Lyme vaccine
is something to stay very far away from. I urge you to do your
own homework and give it great consideration before you or your
loved ones choose to let your doctor provide you this now
‘fast-tracked’, soon to be available Lyme vaccine.
And please – when you do your homework, do NOT fall for the
long-held, propaganda-driven ‘Lyme loonies’, anti-vaxxer’,
‘anti-science’ argument. This is nothing more than a false and
desperate, shall we say childish, position that the powers that
be continue to mount for their own personal benefit driven by
profit and greed, and certainly not public good. Please don’t
fall for these victim blaming, gas lighting, insidious
behaviours. Be smarter than that. It may be too late for the
millions suffering from this life altering illness … but it’s
not too late for you.
Here are the five key reasons why you should NOT get the new
Lyme vaccine:
YOU CANNOT TRUST THE ‘LYME CABAL’
We in the Lyme world have a long list of reasons not to trust
that the medical ‘powers-that-be’ on the subject of Lyme
disease. For forty years, they have been telling us that ‘there
is no such thing as chronic Lyme, that there are no ticks here,
that you couldn’t be this sick, we don’t know what’s wrong with
you but it isn’t Lyme, it’s probably just ‘all in your head’.
Around the world, in 80 countries and on every continent, people
are chronically ill with Lyme disease and most cannot get
treatment. It’s a do-it-yourself disease. Sufferers have to
search far and wide for any type of relief. Most cannot afford
to get out-of-the-box treatments, even if they can find them.
Many die from this disease, too many by their own hand. YET …
for decades now, the Lyme Cabal has been preaching that Lyme
disease is ‘difficult to catch, easy to diagnose and easy to
treat’. Tell the sick and infirm that this is true and see what
happens.
Here is an example of the victim blaming – even worse,
federally-funded victim blaming, using taxpayers hard-earned
dollars:
“Advocacy for Lyme disease has become an increasingly important
part of an antiscience movement that denies both the viral cause
of AIDS and the benefits of vaccines and that supports unproven
(sometimes dangerous) alternative medical treatments. Some
activists portray Lyme disease, a geographically limited
tick-borne infection, as a disease that is insidious,
ubiquitous, difficult to diagnose, and almost incurable; they
also propose that the disease causes mainly non-specific
symptoms that can be treated only with long-term antibiotics and
other unorthodox and unvalidated treatments. Similar to other
antiscience groups, these advocates have created a
pseudoscientific and alternative selection of practitioners,
research, and publications and have coordinated public protests,
accused opponents of both corruption and conspiracy, and spurred
legislative efforts to subvert evidence-based medicine and
peer-reviewed science. The relations and actions of some
activists, medical practitioners, and commercial bodies involved
in Lyme disease advocacy pose a threat to public health.”http://www.thelancet.com/journals/laninf/article/PIIS1473-3099(11)70034-2/abstract
When I had the chance to ask one of the co-authors, Dr. Raymond
Dattwyler why he and his colleagues would write such an article,
his response was “They were bothering us.” Enough said!
So, if in fact as these authors would like you to believe, Lyme
disease is NOT insidious, ubiquitous, difficult to diagnose and
incurable, then why are they bothering to create a vaccine at
all. If Lyme disease is nothing more than a nuisance condition
as some have misclassified it, then why bother? Why inoculate?
Why fast track it? What’s the point? Why the hype? What’s the
rush?
The Centers for Disease Control and Prevention (CDC) -- a
federal agency that conducts and supports health promotion,
prevention and preparedness activities in the United States –
along with the Infectious Diseases Society of America (IDSA)
have been the leading deniers of Lyme disease.
In 2012, when the CDC revised the official case number of new
Lyme disease infections per year from 30,000 to over 300,000 new
Lyme––overnight, those of us in the know understood that they
were getting the public ready to accept this upcoming vaccine
with open arms and a sense of relief. In other words, scare the
public just enough to make them open to wanting this product
without causing mass panic nationwide or worldwide. And without
having to explain their denial to date.
Let it be known that 300,000 cases per year makes Lyme disease
almost twice as common as breast cancer and six times more
common than HIV/AIDS. So why is it that Lyme sufferers receive
far more support and guidance from their fellow Lyme sufferers
on Facebook than in any doctor’s office? Where are the Public
Service Announcements that have not been issued by the
government, the CDC, the IDSA? Why are infectious disease
doctors most notorious for their denial of chronic Lyme disease?
Policy drivers have remained fixated on surveillance of ticks
and the early, non-disseminated, acute stage of Lyme disease,
rather than the millions of Lyme patients who have become
disabled from prolonged exposure to infection and have become
incapacitated. This class of patients have been widely ignored
causing sufferers to have to go it alone, fight the medical
system, experiencing denial of their illness and complete
medical abandonment. Those in the know will tell you that Lyme
disease can and often does become a life altering infection if
not treated immediately. Just ask Duke University Professor Neil
Spector, who required a heart transplant after experiencing four
years of undiagnosed-untreated Lyme disease. Or Dr. Alfred
Miller, a retired Mayo Clinic rheumatologist whose
daughter-in-law was diagnosed with ALS at age 43 which spurred
him to discover links between Lyme and ALS.
Source:https://on-lyme.org/nl/investigators/research/miller
2. THE CRIMINAL ACTS THAT LANDED US HERE
In 1993, the ‘Lyme Cabal’ were in Phase I and II of their OspA
LYMErix vaccine trials.
What is OspA? Outer surface protein A transmitted by two types
of dirty needles––the shed blebs of the spirochete (aka tick
phylum) and the vaccine LYmerix. OspA detonates the immune
system creating the same disease outcome regardless of which way
it’s transmitted.
Make no mistake! The Lyme Cabal knew as early as Phase I trials
that their OspA vaccine would cause the same disease as the
tick-borne illness.
Ergo—their master plan … to create a definition of Lyme disease
that would fit their upcoming vaccine model and thereby get the
vaccine approved and to market.
So …in 1994, the CDC hosted a consensus conference in Dearborn,
MI, along with a dozen labs across the country and together they
falsified the very definition of Lyme disease by eliminating the
neurological, immunosuppressive type which account for 85% of
the cases.
If they conveniently ‘determined’ that the 85% group – those
with an immunosuppression neurological outcome––simply did NOT
exist, then they could claim that their vaccine was 85%
effective. With no immunosuppressive, neurological disease to
find, then the vaccine would be a hit with the remaining 15% who
presented with an arthritic, bad-knee type only. A brilliant
marketing scheme at the expense of millions worldwide for the
years to come.
You see, you CANNOT create a vaccine for an OspA fungal antigen
-- the TRUE definition of chronic Lyme. It can’t be done. And
the OspA vaccination known as LYMErix caused the same disease
from a syringe as it does when you get Lyme disease from a tick
bite. LYMErix victim’s immune systems were destroyed by the
vaccine because OspA is an endotoxin that causes
immunosuppression and subsequent severe neurologic multi-system
disease.
So by narrowing the definition and by claiming that only one
type of the disease (the bad knee arthritic type) exists, then
they could sell a vaccine. Not only that. They were also able to
profit by limiting the number of labs that were sanctioned and
thereby cornering the market on the patents of a variety of tick
borne diseases
3. THE FIRST LYME VACCINE WAS A COMPLETE BUST
In 1998, the FDA approved a new recombinant Lyme vaccine,
LYMErix™.
This OspA vaccine was in the end the very thing, the very
fungal antigen, the very TLR2-agonist (structure equaling
function), that caused the New Great Imitator outcomes of MS,
ALS, Lupus, Chronic Fatigue, Cancer, and more.
LYMErix did not produce antibodies. It is a fungal antigen. It
activates latent herpesviruses, which are basically the main
drivers of the MS and Lupus outcomes. And OspA-induced tolerance
to similar TLR2-agonists causing the ALS and Chronic Fatigue
outcomes (mycoplasma bear OspA-like antigens).
LYMErix vaccine (and the Tuberculosis vaccines) all failed
because they caused immunosuppression, no antibodies, and they
made the victims more susceptible to other infections.
The LYMErix vaccine caused the same disease that us tick bite
sepsis victims know as “Chronic Lyme disease”. It was not taken
off the market due to low sales. The manufacturer SmithKline
Beecham was given an ultimatum in 2002 by the FDA to take it off
the market or they would.
Why would the Lyme cabal purposely create a Lyme vaccine that
is harmful. Well, first and foremost, there is a matter of
patents and profits. Secondly, in order to get the vaccine to
market, they have to show that the vaccine will create
antibodies to produce a certain level of protection. And it is
only when these lipids are exposed (OspA) that the immune system
will see them and cause an immune response. "Falsify some data,
throw out those volunteers, and do whatever you have to to make
the data fit the scientific narrative." says Truthcures activist
Beaux Reliosis. Here is a relatively new scientific report
confirming that this is the case.https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5372817/
The falsified definition of Lyme disease created by the Lyme
Cabal in 1994 followed by the driving force of this criminal act
– their precious LYMErix vaccine – is the entire reason that
millions world-over are suffering from this holy hell and why
infectious disease doctors everywhere will tell you that ‘there
is not such thing as chronic Lyme, it’s no more than a nuisance
disease, it’s all in your head, it’s the celebrities creating
hype and drama that is really the root of the problem.’ Those
celebrities. Shame.
In case you’re interested, here are just a few of the LYMErix
victims’ stories.https://www.fda.gov/ohrms/dockets/ac/01/briefing/3680b2_17.pdf
And this: “A wide range of neurological complications have been
reported via the medical literature and the VAERS system after
vaccination with recombinant outer surface protein A (OspA) of
Borrelia. To explore this issue, 24 patients reporting
neurological adverse events (AE) after vaccination with Lymerix,
out of a group of 94 patients reporting adverse events after
Lymerix vaccination, were examined for causation. Five reports
of cerebral ischemia, two transient Ischemic attacks, five
demyelinating events, two optic neuritis, two reports of
transverse myelitis, and one non-specific demyelinating
condition are evaluated in this paper.”https://www.ncbi.nlm.nih.gov/pubmed/21673416
Bottom line: OspA is Pam3Cys. TLR2/1 fungal endotoxins cannot
be a vaccine.https://www.ncbi.nlm.nih.gov/pubmed/?term=(pam3cys%20or%20pam3*%20or%20p3c)%20and%20(immune%20suppression%20or%20tolerance)
4. AND NOW … A NEW LYME VACCINE IS ABOUT TO BE LAUNCHED
And NOW, The French company Valneva is starting their phase 1
trials here in the United States and in Belgium this year.
Another version of the very same Osp vaccine.
What they fail to mention is that OspA is Pam3Cys. Which leads
to immunosuppression and immune tolerance, reactivated viruses (EBV
and CMV, Herpes) and opportunistic infections, multiple severe
neurologic diseases occurring simultaneously, post-sepsis
syndrome. A B-Cell AIDS destroying the lives of millions.
This new ‘fast-tracked” Lyme vaccine will represent just one
more chapter in this global medical holocaust we know as chronic
Lyme. We're all doing our best to warn YOU––the unsuspecting
public––that you can't vaccinate against an OspA fungal antigen.
It can't be done.
It will do nothing more than make you sick with permanent
immunosuppression … and a life of hell.
Why would BigPharma want to 'sell' yet another OspA vaccine and
risk making people sick? Your guess is as good as mine but we
always know that our best bet is to follow the money. Enough
time has passed since LYMErix. They've worked very hard to blame
the victims for the last fiasco and take no responsibility.
They've created just enough public concern about this so-called
'nuisance disease' that people will be lining up to get it. They
are in a good space right now to bring a new vaccine to market
and make a fortune. As long as they figure out how to mitigate
some of the damage this time, they will walk away with a huge
money-maker, and––yes––with casualties that they can once again
blame on anti-vaxxers, Lyme loonies, and the media.
5. YOU’RE SMARTER THAN THIS!
For the most part, the medical powers that be do NOT have your
best interests at heart. Certainly not when it comes to chronic
Lyme disease. They have too much to hide and nothing to gain by
allowing it to come out from behind the well-placed forty year
shadows. The medical-industrial complex is designed to make
money. Period. It is not designed to make people well or to be
concerned about public good.
Remember this. Do your research. Listen to those suffering. And
think about whether it makes any sense that millions of people
would be faking their illness for some personal gain and allow
their lives to be destroyed for what?
Above all, read, research, ask questions, challenge your
doctors, and think long and hard before you allow yourself or
your loved ones to be forever destroyed by this up-and-coming
Lyme vaccine.
Lori Dennis, MA, RP is a Registered Psychotherapist and the
author of LYME MADNESS, named #1 NEW RELEASE in Immune System
Health on Amazon. LYME MADNESS is available on Amazon. For more
information on Lyme Madness, go to loridennisonline.com.
=======================================================================
22.) SIDE EFFECTS IN YOUNG GIRLS TAKE GARDASIL OUT FROM
JAPANESE MARKET
=======================================================================
Source:ttps://www.tokyotimes.com/side-effects-in-young-girls-take-gardasil-out-from-japanese-market/
Around 2,000 reported side effects after using Gardasil
cervical cancer vaccine have determined Japanese government
officials to withdraw Gardasil from the market in 2013, despite
the vaccine being highly promoted in the United States and now
approved by the European Union.
“Japanese health officials have recorded nearly 2,000 adverse
reactions – hundreds of them serious,” reported Judicial Watch,
the Washington-based corruption watchdog that has been
monitoring the effects – and health costs – of the drug’s use in
the United States for years.
“The alarming reports have led Japan’s government to take
action, suspending recommendation for the controversial vaccine
which is billed as a miracle shot that can prevent certain
strains of cervical cancer caused by Human Papillomavirus (HPV).”
“The U.S. government has taken the opposite approach amid
equally alarming cases of serious side effects. Not only does
the Obama administration continue recommending the vaccine (Gardasil),
it spends large sums of taxpayer dollars promoting it and works
hard to keep details involving its dangers secret.”
The side effects of using Gardasil include seizures, brain
damage, blindness, paralysis, speech problems, pancreatitis and
short-term memory loss, while other patients have died after
taking the vaccine. Gardasil is given to little girls and costs
around $600 per patient.
The organization confirmed that in Japan, the Ministry of
Health, Labor and Welfare warned local governments that the HPV
vaccine should not be recommended amid safety concerns.
Japan’s officials had paid more than $187 million for “urgent
HPV vaccination programs” for girls between 11 and 14 and
visited junior high schools to promote the vaccine.
“Since the government began offering girls HPV shots, 1,968
adverse events were reported, including 358 that were evaluated
as serious by a JMLHW committee. Parents began calling the
country’s health minister and furnishing videos in which girls
who had received the HPV vaccine suffered from walking
disturbances, body tics and seizures. In other cases many girls
injected with the vaccine fell to the floor, injuring their head
or face and some fracturing their jaw or teeth,” Judicial Watch
reported.
The damage payments of nearly $6 million covered only some of
the 200 claims that have been filed to date.
=====================================================================
23.) Vaccines do NOT cause injuries, according to House
Resolution 327
====================================================================
Source:https://www.naturalhealth365.com/vaccines-side-effects-2324.html
(NaturalHealth365) The introduction of House Resolution 327 for
consideration by the U.S. House of Representatives marks an
attempt to take the vaccine injury cover-up to a whole new
level. This action is indicative of the level of dangerous
cluelessness and brainwashing being directed at the American
public, as it relates to the dangers of vaccines.
This new resolution also highlights the influence of money some
of these legislators are likely receiving from big pharma. House
Resolution 327 asserts that there is “no credible evidence” to
show vaccines cause disabling or life-threatening diseases in
healthy adults or children.
This is just one of its numerous demonstrably false claims.
U.S. House members ignore the negative side effects of vaccines
The resolution was introduced by Rep. Adam Schiff (D-Burbank,
CA) and is co-sponsored by at least 33 of his colleagues so far.
These legislators are apparently unfamiliar with even basic
vaccine facts and policy. Vaccine product inserts and the
federal government’s own data list numerous vaccine side effects
and the potential for vaccine injury.
The fact that the vaccine companies themselves warn about the
risk of possible vaccine injury to both children and adults in
otherwise good health should be reason enough to negate this
resolution. However, the individuals behind the draft also seem
to be unaware of the billions of dollars paid out by the vaccine
court to the families of vaccine-injured children.
The vaccine court, or National Vaccine Injury Compensation
Program, has ruled for the plaintiffs in thousands of vaccine
injury claims. This court has paid out over $3.5 billion in
damages thus far since its founding in 1986.
House Resolution 327 calls vaccine side effects and risks
“unfounded”
If vaccines are as safe as Schiff and his colleagues seem to
believe, then why do vaccine companies and doctors seek
liability protection from vaccine injuries? The resolution
itself admits that vaccine recipients must be “monitored for
adverse events” after vaccine shots are given.
House Resolution 327 also has the gall to call efforts toward
vaccine safety and education the dissemination of “unfounded and
debunked” theories about vaccine dangers and their risk to
public health. You can read the entire text of House Resolution
327 here. (Notice how it reads like a press release issued by a
vaccine company).
Full link here:https://www.congress.gov/bill/115th-congress/house-resolution/327/text
Fortunately, House Resolutions do not go directly to the Senate
or President for consideration as a law even if passed. However,
they are considered preferred policy positions and statements of
consensus of the House of Representatives.
Take action: Contact your U.S. House representative
The fact that this resolution contains such outrageously flawed
language and positions is disturbing to say the least. House
Resolution 327 would enshrine demonstrably incorrect information
about vaccines as preferred U.S. House of Representatives
policy.
Numerous elements of the document can be easily refuted just by
referencing the long list of publicly documented vaccine side
effects and injuries.
Anyone who wishes to take action and voice their opposition
regarding this resolution may do so by finding and messaging
their House member directly. Contact information for U.S. House
of Representatives members can be accessed herehttps://www.house.gov
=======================================================================
24.) H. RES. 327
======================================================================
115th CONGRESS
1st Session
Source:https://www.congress.gov/bill/115th-congress/house-resolution/327/text
Recognizing the importance of vaccinations and immunizations in
the United States.
IN THE HOUSE OF REPRESENTATIVES
May 16, 2017
Mr. Schiff (for himself, Mr. Marino, Mr. Cicilline, Mr. Engel,
Ms. Clarke of New York, Mr. Foster, Mr. Lowenthal, Mr. Cohen,
Mr. Langevin, Ms. Michelle Lujan Grisham of New Mexico, Mr.
Cooper, Ms. DeLauro, Mr. Garamendi, Mr. Blumenauer, Ms. DeGette,
Mr. Grijalva, Ms. Eddie Bernice Johnson of Texas, Ms. Eshoo, and
Mr. Dent) submitted the following resolution; which was referred
to the Committee on Energy and Commerce
RESOLUTION
Recognizing the importance of vaccinations and immunizations in
the United States.
Whereas the contributions of Louis Pasteur and Edward Jenner to
the discovery of the principles of vaccination and immunology
are among the most consequential health findings in human
history;
Whereas vaccines have made it possible for the world to have
eradicated smallpox, saving approximately 5 million lives
annually, and for the international community to be on the brink
of eradicating polio and to have saved an estimated 5 million
people from this incurable disease over the past 2 decades,
Whereas vaccines have dramatically reduced the spread of many
more crippling and potentially life-threatening diseases such as
diphtheria, tetanus, measles, mumps, and rubella, and vaccines
prevent the spread of commonly infectious and potentially fatal
diseases such as chickenpox, shingles, influenza, hepatitis A,
hepatitis B, meningococcal disease, pneumococcal, rotavirus, and
whooping cough (pertussis);
Whereas the scientific and medical communities are in
overwhelming consensus that vaccines are both effective and
safe, and the dissemination of unfounded, and debunked, theories
about the dangers of vaccinations pose a great risk to public
health, and scientifically sound education and outreach
campaigns about vaccination and immunization are fundamental for
a well-informed public;
Whereas an estimated 43,000 adults and 300 children die
annually from vaccine-preventable diseases or their
complications in the United States, and the health and
livelihood of young children, seniors, individuals with
immunodeficiency disorders, and those who cannot be vaccinated,
is particularly compromised by communities with low vaccination
rates;
Whereas substantial research has shown that vaccination is a
highly cost-effective form of preventive medicine, and the
Centers for Disease Control and Prevention (CDC) estimates that
vaccinations will save nearly $295 billion in direct costs and
$1.38 trillion in total societal costs in the United States;
Whereas vaccines in the United States undergo exhaustive safety
testing before they are licensed by the Food and Drug
Administration (FDA) and are monitored for adverse events after
health care providers begin administering them to patients;
Whereas there are three post-marketing surveillance systems in
the United States tracking adverse events after vaccination;
Whereas it is estimated that vaccinations will prevent more
than 21 million hospitalizations and 732,000 deaths among
children born in the last 20 years, and that more than 100
million children all over the world are immunized each year and
vaccines have saved an estimated 2.5 million children annually;
Whereas one in five children worldwide still lack access to
even the most basic vaccines and, as a result, an estimated 1.5
million children a year die from vaccine-preventable conditions
such as diarrhea and pneumonia or suffer from permanently
debilitating illnesses;
Whereas a strong investment in medical research to improve
existing vaccines and develop many more life-saving vaccines is
beneficial to all, both at home and abroad, and a robust
immunization infrastructure is essential to the public health
and well-being of the people of the United States by preventing
and isolating outbreaks of contagious diseases where they start;
Whereas encouraging high vaccination rates in the United States
protects our citizens from contracting vaccine-preventable
diseases that are pandemic in countries with low vaccination and
immunization rates;
Whereas routine and up-to-date immunization is the most
effective method available to prevent the infection and
transmission of potentially fatal diseases; and
Whereas the United States has been a leader in promoting
vaccinations around the world through the United States Agency
for International Development, the Centers for Disease Control
and Prevention, Gavi, the Vaccine Alliance, the Global Polio
Eradication Initiative, UNICEF, the World Health Organization,
and a host of other multilateral and nongovernmental
organizations: Now, therefore, be it
Resolved, That the House of Representatives—
(1) commends the international community, global and domestic
health organizations, the private sector, school and community
leaders, and faith-based organizations for their tireless work
and immense contributions to bolstering our global and domestic
health through vaccination;
(2) affirms vaccines and immunizations save lives and are
essential to maintain the public health, and the economic and
national security of the people of the United States;
(3) recognizes that the lack of vaccination can cause a true
public health crisis, and that there is no credible evidence to
show that vaccines cause life-threatening or disabling diseases
in healthy children or adults;
(4) encourages a continued commitment to research to improve
vaccines and to develop new vaccines against other infectious
and fatal diseases; and
(5) urges parents, in consultation with their health care
provider, to follow the scientific evidence and consensus of
medical experts in favor of timely vaccinations to protect their
children and their community.
=======================================================================
25.) Neurological complications of vaccination with outer
surface protein A (OspA).
=======================================================================
Int J Risk Saf Med. 2011;23(2):89-96. doi:
10.3233/JRS-2011-0527.
Marks DH1.
Author information
1
Department of Medicine, Cooper Green Mercy Hospital,
Birmingham, AL, USA. [email protected]
Abstract
A wide range of neurological complications have been reported
via the medical literature and the VAERS system after
vaccination with recombinant outer surface protein A (OspA) of
Borrelia. To explore this issue, 24 patients reporting
neurological adverse events (AE) after vaccination with Lymerix,
out of a group of 94 patients reporting adverse events after
Lymerix vaccination, were examined for causation. Five reports
of cerebral ischemia, two transient Ischemic attacks, five
demyelinating events, two optic neuritis, two reports of
transverse myelitis, and one non-specific demyelinating
condition are evaluated in this paper. Caution is raised on not
actively looking for neurologic AE, and for not considering
causation when the incidence rate is too low to raise a
calculable difference to natural occurence.
=======================================================================
26.) Spirochetal Lipoproteins and Immune Evasion
=======================================================================
Front Immunol. 2017; 8: 364.
Published online 2017 Mar 29. doi: 10.3389/fimmu.2017.00364
Alexei Christodoulides,1 Ani Boyadjian,1 and Theodoros
Kelesidis1,*
Abstract
Spirochetes are a major threat to public health. However, the
exact pathogenesis of spirochetal diseases remains unclear.
Spirochetes express lipoproteins that often determine the cross
talk between the host and spirochetes. Lipoproteins are
pro-inflammatory, modulatory of immune responses, and enable the
spirochetes to evade the immune system. In this article, we
review the modulatory effects of spirochetal lipoproteins
related to immune evasion. Understanding lipoprotein-induced
immunomodulation will aid in elucidating innate pathogenesis
processes and subsequent adaptive mechanisms potentially
relevant to spirochetal disease vaccine development and
treatment.
======================================================================
27.) Does a New Hepatitis Vaccine Cause Heart Attacks?
=====================================================================
Source:https://www.medpagetoday.com/blogs/revolutionandrevelation/67019
by Milton Packer
August 02, 2017
The FDA has a really important question and wants your advice.
This is not a fairy tale. This is a real-life story.
Hepatitis B is a serious disease. A company (Dynavax) has a new
hepatitis vaccine that induces hepatitis antibodies more
vigorously than existing vaccines and does so after 2 doses
(instead of the usual 3). The vaccine works through a unique
adjuvant. The serological advantages of the Dynavax vaccine were
demonstrated in a randomized trial of >8000 patients; about 5600
people received the new vaccine and about 2800 people received
the existing standard.
Why does the FDA need your help?
In the trial, an acute myocardial infarction occurred in 14
people in the Dynavax group, but in only one person receiving
the conventional vaccine. The events were confirmed by
adjudication. Since the Dynavax group was twice as large, the
risk of acute myocardial infarction in the trial was seven times
greater with the new vaccine. The FDA wants to know if the new
vaccine should be approved for use in millions of people.
What do you say? What recommendation would you make?
If you think this is just hypothetical, think again. On July
28, 2017, the FDA convened a public advisory committee meeting
to consider this exact question. The members of the committee
consisted primarily of experts in infectious diseases and
immunology. I was the only cardiologist on the committee.
If the 14:1 imbalance was due to the play of chance, then the
issue of myocardial infarction risk was spurious, and the
vaccine should be approved. However, if the 14:1 imbalance
reflected a real increase in cardiovascular risk, then approval
of Dynavax vaccine would be problematic.
Was it biologically plausible for the new vaccine to cause
heart attacks?
The new adjuvant in the vaccine caused an inflammatory response
(of uncertain duration), and inflammation is an important cause
of rupture of atherosclerotic plaques. So a causal linkage was
not out of the question.
Was the imbalance in myocardial infarctions due to the play of
chance?
That was a good question, but it was impossible to know. Many
might think that calculation of a P value would help, but it
wouldn't. P values have a place in clinical trials, but not when
the number of events is so small and the number of comparisons
is so great. So no one asked for or showed any P values during
the meeting. Everyone agreed that statistics could not resolve
the uncertainty.
If you wanted to know if the 14:1 imbalance represented a real
risk, you needed more information. You needed comparative data
in 50,000 people. The fastest way of obtaining that evidence was
through a post-marketing trial. But a post-marketing trial was
possible only if the vaccine was approved for public use.
So what recommendation would you have made to the FDA?
The FDA asked the committee if there was reasonable evidence
that the vaccine was safe. On July 28, the committee vote 12-1
(with 3 abstentions) in favor of the safety of the new vaccine.
I was one of the three abstentions. Most of the committee
believed that the vaccine's serological advantages outweighed
the uncertainty, but the vote is non-binding. The FDA will
decide on the new vaccine by August 10.
Why did I abstain? Based on the available data, it was
impossible for anyone to know if the imbalance in myocardial
infarctions was real or spurious. So although the question was
fascinating and the discussion was terrific, my vote wasn't that
complicated.
There is a simple rule in life: if you don't know, you should
say that you don't know.
=======================================================================
28.) FDA extends review on Dynavax's Heplisav-B, but management
remains confident
=======================================================================
source:http://www.fiercepharma.com/vaccines/dynavax-shares-roller-coaster-as-fda-requires-heplisav-b-postmarketing-details
The fate of hepatitis B vaccine candidate Heplisav-B has sent
developer Dynavax’s shares, and its investors, on a wild ride in
recent days. A request by the FDA for more details of the
vaccine’s postmarketing surveillance plan sent the company's
stock slightly downward, but both management and analysts said
they remain confident in approval.
Given the closeness of the FDA's action date for the vaccine on
August 10, Dynavax said that after discussion with the agency
they have agreed to extend the review date for up to three
months to finalize details of the postmarketing study.
The agency’s request mainly centers on ensuring accurate,
timely collection of real-world safety data from Heplisav-B use.
During an analyst call Thursday afternoon, Dynavax CEO Eddie
Gray stressed that the FDA’s request is “wholly consistent with”
what the agency's expert panel had expressed earlier in the
week. During that panel, experts voted 12 to 1 in favor of the
shot’s safety profile, a result that sent Dynavax’s stock to a
one-year high. But the panel also raised concerns about the
proposed design of the surveillance program.
RELATED: Heplisav-B approval ‘highly probable’ as FDA Advisory
Committee favors Dynavax on third try
“This delay … does not in any way impact our confidence that
the vaccine should be approved or our internal launch plans and
timeline,” Gray said during the call.
Perhaps because the issue was already well expressed during the
FDA panel discussion, Dynavax's share price is gradually
recovering from a slight slip Friday. At least one analyst
shared Dynavax’s optimism.
“[W]e don’t view this delay as fundamentally changing the
narrative, and continue to see approval as a matter of when, not
if,” RBC Capital Markets analyst Matthew Eckler wrote in a note
to investors. “The recent positive FDA AdCom was a key derisking
event for Heplisav-B, and given the vaccine’s superior profile,
it has the potential to capture significant market share, as
well as drive expansion.”
Eckler previously projected that the vaccine could reach peak
U.S. sales of $290 million in 2026.
Under the current plan, Dynavax is aiming to launch the shot, a
challenger to GlaxoSmithKline’s Engerix-B, in the U.S. in early
2018. It's proposing to conduct the phase 4 study in
collaboration with Kaiser Permanente Northern California to
monitor medical records for 20,000 Heplisav-B recipients against
20,000 others who receive the GSK shot during a 12-month
follow-up. It's likely that cardiac incidents, for which an
imbalance was seen in a phase 3 study, will be the main focus.
Gray added that the company is on track to present to the CDC’s
Advisory Committee on Immunization Practices at their meeting in
late October, even if it’s before the Nov. 10 FDA decision
deadline.
=====================================================================
29.) Can Hib Vaccine Cause Injury?
=====================================================================
Source:http://www.nvic.org/vaccines-and-diseases/hib/hib-vaccine-injury.aspx
Mild side effects such as redness, warmth, or swelling where
the shot was given have been reported in connection with
administration of Hib vaccines. Fever over 101 degrees F may
occur, and can last two to three days. Sysemic reactions include
irritability and lethargy. However, more severe reactions have
also been reported in both clinical trials with all of the
vaccines as well as to the Vaccine Adverse Events Reporting
System (VAERS).
According to MedAlerts.org (a searchable VAERS database) as of
June 2012, there have been 12,140 serious adverse events
reported to VAERS in connection with all Hib vaccines combined.
Most of this number were children under age 3 (11,278). Serious
reactions included deaths (471) and such things as anaphylactic
reaction, asthma, pneumonia, convulsions, noninfectious
encephalitis, acute pancreatitis, peripheral neuropathy,
Guillain-Barre syndrome, sepsis, seizures, cerebral edema.
In clinical trials the severity of adverse reactions varied
depending on which vaccine was given, and which other vaccines
were given with them at the same time. Some of the events
reported by the manufacturers included:
ActHIB — tenderness, erythema, induration, fever, irritability,
drowsiness, anorexia, diarrhea, vomiting; when combined with DTP
vaccine by reconstitution, adverse reactions included:
tenderness, erythema, induration, fever, irritability,
drowsiness, anorexia, diarrhea, persistent crying, and one
hypotonic/hypresponsive episode (which is consistent with the
HHE rate observed with DTP vaccination alone)
Hiberix — when co-administered with DTaP-HBV-IPV: redness, pain
and swelling at injection site, fever, fussiness, loss of
appetite, restlessness, sleepiness, diarrhea, vomiting;
post-marketing reported adverse events included extensive
swelling of the vaccinated limb, anaphylactic reactions,
angioedema, convulsions, hypotonic-hyporesponsive episode,
syncope, apnea, rash, urticarial, somnolence.
PedvaxHIB — adverse events during clinical trials included
irritability, sleepiness, injection siter pain/soreness,
erythema, swelling induration, unusual high-pitched crying,
prolonged crying (more than 4 hours), diarrhea, vomiting,
crying, pain, otitis media, rash, and upper respiratory
infection; potential adverse events may include early onset of
Hib disease and Guillain-Barre syndrome; in post-marketing,
reported adverse events included lymphadenopathy, angioedema,
febrile seizures and injection site abscess.
Comvax — (children in clinical trials were monitored five days)
with the most frequently cited events being mild, transient
signs and symptoms of inflammation at the injection site,
pain/soreness, erythema, swelling, induration, somnolence,
irritability, anorexia, vomiting, otitis media, fever, diarrhea,
upper respiratory infection, rash, rhinorrhea, respiratory
congestion, cough, candidiasis; in post-marketing: anaplylaxis,
angioedema, urticarial, erythema multiforme, thrombocytopenia,
seizure, febrile seizures, lymphadenopathy, pruritus, arthralgia,
dyspnea, tachycardia, syncope, elevation of liver enzymes,
increased erythrocyte sedimentation rate, arthritis, Bell’s
Palsy, Guillain-Barre syndrome, agitation, Stevens-Johnson
syndrome, alopecia, conjunctivitis, visual disturbances.
Pentacel — redness, swelling, tenderness at injection site,
increase in arm circumference (dose 4), fever, lethargy,
inconsolable crying, fussiness, irritability, hypotonic
hyporesponsive episodes, seizures, febrile seizures,
bronchiolitis, dehydration, pneumonia, gastroenteritis, asthma,
pneumonia, encephalopathy, and four deaths attributed to
asphyxia due to suffocation, head trauma, Sudden Infant Death
syndrome (SIDS), and neuroblastoma; in post-marketing, reported
adverse events included cyanosis, vomiting, diarrhea, extensive
swelling of injected limb including swelling that involved
adjacent joints, invasive Hib disease (classified as vaccine
failure), rash, urticarial, meningitis, rhinitis, viral
infection, decreased appetitie, somnolence, HHE, depressed level
of consciousness, screaming, apnea, cough, erythema, skin
discoloroation, and pallor.
MenHibrix — redness, swelling and pain at injection site,
irritability, drowsiness, loss of appetitie, fever, and syncope.
For more severe reactions such as nervous system disorders and
other serious events, the manufacturer referred to reactions
reported by the use of Hiberix rather than its own vaccine. It
is not known whether MenHibrix can cause fetal harm when
administered to a pregnant woman or whether it can affect
reproduction capacity. 1
Besides the deaths reported in the clinical trials, deaths have
been recorded in post-marketing reports as well. In fact, an
open letter to Dr. Margaret Chan, director general of the World
Health Organization, in March 2012 calls attention to the deaths
the author says are connected with the pentavalent (DPT + Hib +
Hepatitis B vaccine in India, Sri Lanka, Bhutan, and Pakistan.2
The authors add that the cause of the problem is unrelated to
the brand or manufacturer or lot of the vaccine:
“It appears to be a form of ‘hypersensitivity reaction’ as
described in the post mortem report on one of the children in
Kerala. The vaccine can be administered to many patients without
problems and there is no available method at present to predict
which infant will react adversely.”
In a pediatric safety and use review of Pentacel in February
2010 the U.S. FDA discussed reported adverse reactions
attributed to the vaccine in post-marketing, noting that
Pentacel is now marketed in 11 countries, and there have been no
safety-related labeling revisions. At the meeting, the FDA
reported that 775 adverse reactions, 177 of which were
considered serious including 26 deaths, had been reported to
VAERS in connection with Pentacel between June 20, 2008 (when
Pentacel was licensed) and October 31, 2009. The deaths were
attributed to SIDS (12 cases), congenital/genetic conditions,
respiratory infections, positional asphyxia, anoxic
encephalopathy, cardiac arrest of undetermined etiology, dilated
and hypertrophic cardiomyopathy, two deaths of undetermined
cause, one death with no records obtainable, and two deaths with
information pending.
The FDA said the SIDS reports “do not raise any concerns about
a causal relationship with Pentacel.” The FDA also quoted the
Institue of Medicine, which reviewed the data, and the IOM said:
“The evidence favors rejection of a causal relationship between
exposure to multiple vaccines and SIDS.” The FDA noted that “the
sponsor is voluntarily conducting a descriptive, epidemiological
safety surveillance study that will include at least 10,000
Pentacel recipients” with comparison groups of infants who
receive other DTaP-containing products.3
Antigenuria has been reported after receipt of purified
capsular polysaccharide H. influenza b vaccine. In one reported
case, a 27-month-old child developed asceptic meningitis two
days after getting the vaccine. Tests revealed that antigenuria
was secondary to the vaccination.4,5,6None of the vaccines have
been evaluated for carcinogenic or mutagenic potential, or
potential to impair fertility.
=======================================================================
===================================================================
DATA-MEDICOS/DERMAGIC-EXPRESS No 19-(203) 22/10/2.017 DR. JOSE LAPENTA R.
===================================================================
Produced
by Dr. Jose Lapenta R. Dermatologist 2.017
Maracay Estado Aragua Venezuela 2.017
Telf: 0416-6401045- 02432327287-02432328571
loading...
|